BONSPRI Solution for injection Ref.[50516] Active ingredients: Ofatumumab
Source: Health Products Regulatory Authority (ZA) Revision Year: 2021 Publisher: Novartis South Africa (Pty) Ltd., Magwa Crescent West, Waterfall City, Jukskei View, Johannesburg, 2090
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: selective immunosuppressants
ATC code: not yet assigned
Mechanism of action
B cells play an important role in MS pathogenesis due to production of pro-inflammatory cytokines, release of auto-reactive antibodies and activation of pathogenic T cells. Ofatumumab is a fully human anti-CD20 monoclonal antibody (IgG1). It binds to a distinct epitope encompassing both the small and large extracellular loops of the CD20 molecule, giving rise to a slow off-rate and high binding affinity. The CD20 molecule is a transmembrane phosphoprotein expressed on B lymphocytes from the pre-B to mature B lymphocyte stage. The CD20 molecule is also expressed on a small fraction of activated T cells.
The binding of ofatumumab to CD20 induces lysis of CD20+ B cells primarily through complement-dependent cytotoxicity (CDC) and, to a lesser extent, through antibody-dependent cell-mediated cytotoxicity (ADCC). Ofatumumab has also been shown to induce cell lysis in both high and low CD20 expressing cells. CD20-expressing T cells are also depleted by ofatumumab.
Pharmacodynamic effects
B-cell depletion
In the RMS phase III studies, ofatumumab 20 mg every 4 weeks, after an initial dose regimen of 20 mg on days 1, 7, and 14, resulted in a rapid and sustained reduction of B cells to below the lower limit of normal as early as two weeks after treatment initiation, and sustained for as long as 120 weeks while on treatment.
Similar results were observed in a study of bioequivalence using the same dosing regimen as in the phase III studies. Before initiation of the maintenance phase starting at week 4, total B-cell levels of <10 cells/µl were reached in 94% of patients, increasing to 98% of patients at week 12.
B-cell repletion
Data from RMS clinical studies indicate B-cell recoveries over the LLN in at least 50% of patients in 24 to 36 weeks post treatment discontinuation. Modelling and simulation for B-cell repletion corroborate this data, predicting median time to B-cell recovery of 40 weeks post treatment discontinuation.
Immunogenicity
As a fully human monoclonal antibody, ofatumumab has a low potential of inducing anti-drug antibodies (ADA). In RMS phase III studies, the overall incidence of ADAs was very low: treatment-induced ADA were detected in 2 of 914 ofatumumab-treated patients and no patients with treatment enhancing or neutralising ADA were identified. There was no impact of positive ADA titers on PK, safety profile or B-cell kinetics in any patient.
Clinical efficacy and safety
The efficacy and safety of BONSPRI were evaluated in two randomised, double-blind, active-controlled phase III pivotal studies of identical design (G2301 [ASCLEPIOS I] and G2302 [ASCLEPIOS II]) in patients with relapsing forms of MS (RMS) aged 18 to 55 years, a disability status at screening with an Expanded Disability Status Scale (EDSS) score from 0 to 5.5, and who had experienced at least one documented relapse during the previous year or two relapses during the previous two years or positive gadolinium (Gd)-enhancing MRI scan during the previous year.
In the two studies, 927 and 955 patients with RMS, respectively, were randomised 1:1 to receive either ofatumumab 20 mg subcutaneous injections every 4 weeks starting at week 4 after an initial dosing regimen of three weekly 20 mg doses in the first 14 days (on days 1, 7 and 14) or teriflunomide 14 mg capsules orally once daily. Patients also received matching placebo corresponding to the other treatment arm to ensure blinding (double-dummy design).
The treatment duration for individual patients was variable based on when the end of study criteria were met. Across both studies, the median treatment duration was 85 weeks, 33,0% of patients in the ofatumumab group vs 23,2% of patients in the teriflunomide group were treated more than 96 weeks.
Demographics and baseline characteristics were well-balanced across treatment arms and both studies (see Table 2). Mean age was 38 years, mean disease duration was 8,2 years since onset of first symptom, and mean EDSS score was 2.9; 40% of patients had not been previously treated with a disease-modifying therapy (DMT) and 40% had gadolinium (Gd)-enhancing T1 lesions on their baseline MRI scan.
The primary efficacy endpoint of both studies was the annualised rate of confirmed relapses (ARR) based on EDSS. Key secondary efficacy endpoints included the time to disability worsening on EDSS (confirmed at 3 months and 6 months), defined as an increase in EDSS of ≥1.5, ≥1, or ≥0.5 in patients with a baseline EDSS of 0, 1 to 5, or ≥5.5, respectively. Further key secondary endpoints were the time to disability improvement on EDSS (confirmed at 6 months), the number of Gd-enhancing T1 lesions per MRI scan, the annualised rate of new or enlarging T2 lesions, the neurofilament light chain (NfL) concentration in serum and the rate of brain volume loss (BVL). Disability-related key secondary endpoints were evaluated in a meta-analysis of combined data from studies G3201 and G2302, as defined in the study protocols.
Table 2. Demographics and baseline characteristics:
| Characteristics | Study G2301 (ASCLEPIOS I) | Study G2302 (ASCLEPIOS II) | ||
|---|---|---|---|---|
| Ofatumumab (N=465) | Teriflunomide (N=462) | Ofatumumab (N=481) | Teriflunomide (N=474) | |
| Mean age (years) | 38.9 | 37.8 | 38.0 | 38.2 |
| Age range (years) | 19-55 | 18-55 | 18-55 | 18-55 |
| Female (%) | 68.4 | 68.6 | 66.3 | 67.3 |
| Mean/Median duration of MS since first symptoms (years) | 8.36 / 6.41 | 8.18 / 6.69 | 8.20 / 5.70 | 8.19 / 6.30 |
| Mean/Median duration of MS since diagnosis (years) | 5.77 / 3.94 | 5.64 / 3.49 | 5.59 / 3.15 | 5.48 / 3.10 |
| Previously treated with DMTs (%) | 58.9 | 60.6 | 59.5 | 61.8 |
| Number of relapses in last 12 months | 1.2 | 1.3 | 1.3 | 1.3 |
| Mean/Median EDSS score | 2.97 / 3.00 | 2.94 / 3.00 | 2.90 / 3.00 | 2.86 / 2.50 |
| Mean total T2 lesion volume (cm³) | 13.2 | 13.1 | 14.3 | 12.0\ |
| Patients free of Gd+ T1 lesions (%) | 62.6 | 63.4 | 56.1 | 61.4 |
| Number of Gd+ T1 lesions (mean) | 1.7 | 1.2 | 1.6 | 1.5 |
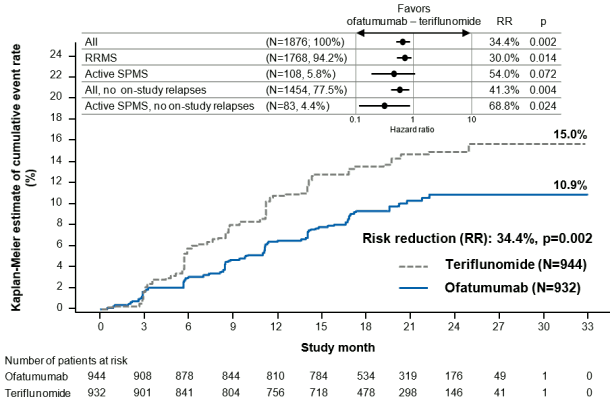
The efficacy results for both studies are summarised in Table 3, Figure 1 and Figure 3.
In both phase III studies (G2301 and G2302), BONSPRI demonstrated a significant reduction in the annualised relapse rate of 50,5% and 58,5%, respectively (both p<0.001) compared to teriflunomide.
The pre-specified meta-analysis of combined data showed that BONSPRI significantly reduced the risk of 3-month confirmed disability worsening (CDW) (risk reduction = 34,4%, p=0.002) and 6-month CDW (risk reduction = 32,5%, p=0.012) compared to teriflunomide (see Figure 1).
BONSPRI significantly reduced the number of Gd-enhancing T1 lesions and the rate of new or enlarging T2 lesions by 95,9% and 83,6%, respectively (both studies combined).
Efficacy results were consistent across the two phase III studies (G2301 and G2302) and across subgroups defined based on gender, age, prior MS therapy, baseline and on-study relapse activity, baseline MRI disease activity, baseline EDSS, and RRMS/SPMS diagnosis.
Table 3. Overview of results from phase III studies in RMS:
| Endpoints | Study G2301 (ASCLEPIOS I) | Study G2302 (ASCLEPIOS II) | ||
|---|---|---|---|---|
| Ofatumumab 20 mg (n=465) | Teriflunomide 14 mg (N=462) | Ofatumumab 20 mg (N=481) | Teriflunomide 14 mg (N=474) | |
| Endpoints based on separate studies | ||||
| Annualised relapse rate (ARR) (primary endpoint)1 | 0.11 | 0.22 | 0.10 | 0.25 |
| Rate reduction | 50,5% (p <0.001) | 58,5% (p <0.001) | ||
| Mean number of T1 Gd-enhancing lesions per MRI scan | 0.0115 | 0.4523 | 0.0317 | 0.5141 |
| Relative reduction | 97,5% (p <0.001) | 93,8% (p <0.001) | ||
| Number of new or enlarging T2 lesions | 0.72 | 4.00 | 0.64 | 4.15 |
| Relative reduction | 82,0% (p <0.001) | 84,5% (p <0.001) | ||
| NfL2 at month 3 (pg/ml) | 8.80 | 9.41 | 8.92 | 10.02 |
| Relative reduction | 7% (p=0.011) | 11% (p <0.001) | ||
| NfL2 at month 12 (pg/ml) | 7.02 | 9.62 | 7.06 | 9.53 |
| Relative reduction | 27% (p <0.001) | 26% (p <0.001) | ||
| NfL2 at month 24 (pg/ml) | 6.90 | 8.99 | 6.80 | 8.99 |
| Relative reduction | 23% (p <0.001) | 24% (p <0.001) | ||
| Endpoints based on pre-specified meta-analyses | ||||
| Proportion of patients with 3-month confirmed disability worsening3 | 10,9% ofatumumab vs. 15,0% teriflunomide | |||
| Risk reduction (meta-analysis) | 34,4% (p=0.002) | |||
| Proportion of patients with 6-month confirmed disability worsening4 | 8,1% ofatumumab vs. 12,0% teriflunomide | |||
| Risk reduction (meta-analysis) | 32,5% (p=0.012) | |||
1 Confirmed relapses (accompanied by a clinically relevant change in the EDSS).
2 In serum
3 Defined as at least a 1 point increase from baseline in EDSS sustained for 3 months (0.5 point increase for patients with baseline EDSS of 5.5 or more and 1.5 point increase for patients with baseline EDSS of 0).
4 Defined as at least a 1 point increase from baseline in EDSS sustained for 6 months (0.5 point increase for patients with baseline EDSS of 5.5 or more and 1.5 point increase for patients with baseline EDSS of 0).
Figure 1. Time to first 3-month CDW by treatment (G2301 and G2302 combined, full analysis set) and subgroups:
Elevated levels of neurofilament light chain (NfL) in serum are a specific marker of neuronal injury. In both phase III studies (G2301 and G2302), BONSPRI significantly reduced NfL concentrations at month 3 (p=0.011) and in all post-baseline visits compared with teriflunomide (see Table 3).
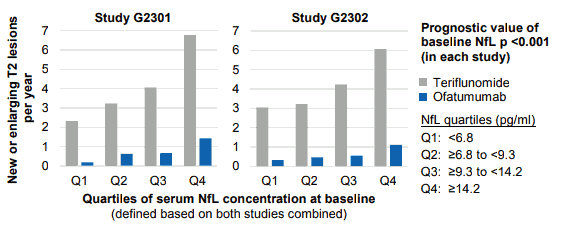
Furthermore, in both studies higher NfL concentrations at baseline were correlated with higher number of new or enlarging T2 lesions by the end of study, i.e. NfL had prognostic value (p<0.001) for on-study lesion formation (see Figure 2). BONSPRI reduced the number of on-study lesions, irrespective of the baseline NfL level.
Figure 2. Number of new or enlarging T2 lesions per year (at the end of the study relative to baseline), by NfL baseline quartiles:
Figure 3. Annualised relapse rates (G2301 and G2302 combined, full analysis set) by subgroup:
5.2. Pharmacokinetic properties
Ofatumumab exhibits a long half-life and low volume of distribution similar to that of other monoclonal antibodies. Ofatumumab is eliminated through a non-linear target-mediated route as well as a target-independent route mediated by non-specific endocytosis followed by intracellular catabolism. Higher baseline B-cell count results in greater component of target-mediated elimination clearance and shorter ofatumumab half-life at the start of therapy. Subsequent ofatumumab dosing leads to potent depletion of B cells resulting in reduced overall clearance.
Absorption
A monthly subcutaneous dose of 20 mg leads to a mean AUCtau of 483 µg·h/ml and a mean Cmax of 1,43 µg/ml at steady state.
After subcutaneous administration, ofatumumab is believed to be predominantly absorbed via the lymphatic system similarly to other therapeutic monoclonal antibodies.
Distribution
Overall, ofatumumab steady-state volume of distribution (Vss) values were low (3 to 8 litres), consistent with other monoclonal antibodies. Following two intravenous infusions of ofatumumab (100, 300 or 700 mg in either the first or second treatment period) in patients with relapsing remitting multiple sclerosis (RRMS), geometric mean Vss after the second infusion of ofatumumab ranged from 2,15 to 2,74 litres. Based on pharmacokinetic modelling of the data from the studies using subcutaneous administration and repeated 20 mg doses, a central volume (Vc) of 2,8 litres was estimated.
Biotransformation / metabolism
Ofatumumab is a protein for which the expected metabolic pathway is degradation to small peptides and amino acids by ubiquitous proteolytic enzymes.
Elimination
Ofatumumab is eliminated in two ways: a target-independent route as with other IgG molecules and a target-mediated route that is related to binding to B cells. Following a single subcutaneous administration of 30 to 100 mg in patients with rheumatoid arthritis, ofatumumab geometric mean elimination half-life values ranged from 5,2 to 6,8 days. Based on pharmacokinetic modelling of the data from the studies using subcutaneous administration and repeated 20 mg doses, an approximate half-life of ofatumumab of 14,9 days in men and 17,1 days in women was estimated.
Linearity / non-linearity
Ofatumumab had non-linear pharmacokinetics related to its decreasing clearance over time.
Special populations
Adults over 55 years old
There are no dedicated pharmacokinetic studies of ofatumumab in patients over 55 years old due to limited clinical experience (see section 4.2).
Paediatric population
No studies have been conducted to investigate the pharmacokinetics of ofatumumab in paediatric patients below the age of 18 years.
Gender
Gender had a modest (12%) effect on ofatumumab central volume of distribution in a cross-study population analysis, with higher Cmax and AUC values observed in female patients (48% of the patients in this analysis were male and 52% were female); these effects are not considered clinically relevant, and no dose adjustment is recommended.
Renal impairment
Ofatumumab is not excreted via urine; therefore, it is not expected that patients with renal impairment require dose modification.
Hepatic impairment
Since hepatic metabolism of monoclonal antibodies such as ofatumumab is negligible, hepatic impairment is not expected to impact its pharmacokinetics. Therefore, it is not expected that patients with hepatic impairment require dose modification.
5.3. Preclinical safety data
Non-clinical data revealed no special hazard for humans based on conventional studies of repeated dose toxicity including safety pharmacology endpoints.
In all pivotal repeat-dose toxicity studies, the highest dose of 100 mg/kg ofatumumab was defined as the no observed adverse effect level (NOAEL). This corresponds to safety margins of at least 110-fold when compared with the clinical exposure at the therapeutic dose of 20 mg monthly.
Neither carcinogenicity nor mutagenicity studies have been conducted with ofatumumab. As an antibody, ofatumumab is not expected to interact directly with DNA.
The embryo-foetal development (EFD) and the enhanced pre/post-natal development (ePPND) studies in monkeys showed that exposure to ofatumumab given intravenously during gestation caused no maternal toxicity, no teratogenicity, and no adverse effects on embryo-foetal and pre/post-natal development. The NOAEL for these parameters leads to AUC-based safety margins of at least 160-fold when compared with human exposure at the therapeutic dose of 20 mg monthly.
In these studies, ofatumumab was detected in the blood of the foetuses and infants, confirming placental transfer and foetal exposure to ofatumumab persisting post-natally (long half-life of the monoclonal antibody). Exposure to ofatumumab during gestation led to the expected depletion of CD20+ B cells in maternal animals and their foetuses and infants, along with a reduced spleen weight (without histological correlate) in foetuses and a reduced humoral immune response to keyhole limpet haemocyanin (KLH) in infants at high doses. All these changes were reversible during the 6-month post-natal period. In infants, early post-natal mortality was observed at a dose 160 times higher than the therapeutic dose (on AUC basis) and was likely due to potential infections secondary to immunomodulation. The NOAEL related to the pharmacological activity of ofatumumab in infants of the ePPND study leads to an AUC-based safety margin of at least 22-fold when maternal exposure at the NOAEL is compared with human exposure at the therapeutic dose of 20 mg monthly.
In a dedicated monkey fertility study, male and female fertility endpoints were unaffected. The NOEL-related exposure is at least 260 times higher than the human exposure at the therapeutic dose of 20 mg monthly in terms of AUC.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.