DAURISMO Film-coated tablet Ref.[10664] Active ingredients: Glasdegib
Source: European Medicines Agency (EU) Revision Year: 2021 Publisher: Pfizer Europe MA EEIG, Boulevard de la Plaine 17, 1050 Bruxelles, Belgium
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: antineoplastic agents, other antineoplastic agents
ATC code: L01XX63
Mechanism of action
Glasdegib is an inhibitor of the Hedgehog (Hh) signal transduction pathway that binds to Smoothened (SMO), a transmembrane protein, leading to decreased Glioma-Associated Oncogene (GLI) transcription factor activity and downstream pathway signalling. Hh pathway signalling is required for maintaining a leukaemic stem cell (LSC) population thus, glasdegib binding to and inhibiting SMO reduces GLI1 levels in AML cells and the leukaemic initiating potential of AML cells. Hh pathway signalling is also implicated in resistance to chemotherapy and targeted therapy. In a preclinical model of AML, glasdegib in combination with low-dose cytarabine inhibited increases in tumour size to a greater extent than glasdegib or low-dose cytarabine alone. However, mechanism of action of the combination is not fully understood.
Cardiac electrophysiology
Heart rate corrected QT (QTc) interval prolongation has been observed in patients treated with Daurismo at a supratherapeutic dose of >270 mg. The effect of glasdegib administration on corrected QT interval was evaluated in a randomised, single-dose, double-blind, 4-way crossover, placebo- and open-label moxifloxacin controlled study in 36 healthy subjects. At therapeutic plasma concentrations (achieved with a 150 mg single dose), the largest, placebo and baseline-adjusted corrected QT interval change was 8.03 msec (90% CI: 5.85, 10.22 msec). At approximately twice the therapeutic concentration (supratherapeutic, achieved with a 300 mg single dose), the QTc change was 13.43 msec (95% CI: 11.25, 15.61 msec). Moxifloxacin (400 mg), used as a positive control, showed a mean QTc change from baseline of 13.87 msec. None of the subjects met categorical criterion of absolute corrected QT interval of ≥480 msec or increase from baseline in corrected QT interval ≥30 msec after receiving any treatment. None of the ECG abnormalities were considered clinically significant or reported as adverse events by the investigator (see section 4.4).
Additionally, serial, triplicate ECGs were collected following a single and multiple dosing to evaluate the effect of single agent glasdegib on the corrected QT interval in 70 patients with advanced cancer (5 mg to 640 mg once daily). Based on the exposure-response analysis, the estimated mean change from baseline in QTc was 5.30 msec (95% CI: 4.40, 6.24 msec) at the mean observed Cmax at steady state following administration at the recommended 100 mg once daily dose of glasdegib.
Clinical efficacy and safety
Daurismo in combination with low-dose cytarabine was investigated in a multicentre, randomised, open-label Phase 2 study (Study 1) in a total of 132 patients, which included 116 patients with previously untreated de novo or secondary AML who were not eligible to receive intensive chemotherapy as defined by meeting at least one of the following criteria: a) age >75 years, b) severe cardiac disease, c) baseline Eastern Cooperative Oncology Group (ECOG) performance status of 2, or d) baseline serum creatinine >1.3 mg/dL. Patients were randomised 2:1 to receive Daurismo (100 mg orally once daily) with low-dose cytarabine (20 mg SC twice daily on days 1 to 10 of the 28-day cycle) (n=78) or low-dose cytarabine alone (n=38) in 28-day cycles until disease progression or unacceptable toxicity. Patients were stratified at randomisation by prognostic risk factor (good/intermediate or poor) based on cytogenetics.
The baseline demographic and disease characteristics are shown in Table 7. The two treatment arms were generally balanced with respect to the baseline demographics and disease characteristics. Across both arms, 40% of the AML patients had poor cytogenetic risk and 60% had good/intermediate cytogenetic risk.
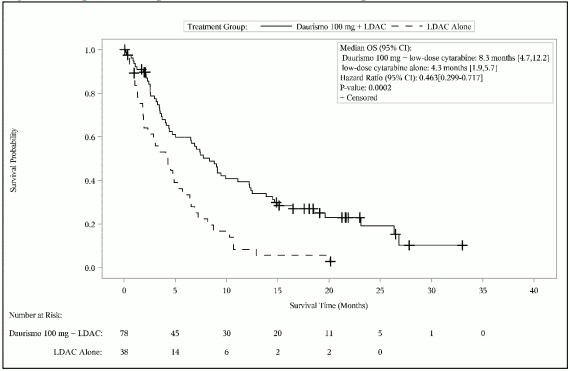
Efficacy was established by an improvement in overall survival (OS defined from the date of randomisation to death of any cause) in the Daurismo with low-dose cytarabine arm, compared to low-dose cytarabine alone. After a median follow-up of approximately 20 months with 81% deaths observed, the Daurismo with low-dose cytarabine arm was superior to low-dose cytarabine alone in AML patients (Figure 1). The efficacy results are shown in Table 8.
Table 7. Baseline demographic and disease characteristics in patients with AML:
| Demographic and disease characteristics | Daurismo with low-dose cytarabine (N=78) | Low-dose cytarabine alone (N=38) |
|---|---|---|
| Demographics | ||
| Age | ||
| Median (Min, Max) (Years) | 77 (64, 92) | 76 (58, 83) |
| ≥75 years N (%) | 48 (62) | 23 (61) |
| Sex, N (%) | ||
| Male | 59 (76) | 23 (61) |
| Female | 19 (24) | 15 (39) |
| Race, N (%) | ||
| White | 75 (96) | 38 (100) |
| Black or African American | 1 (1) | 0 (0) |
| Asian | 2 (3) | 0 (0) |
| Disease characteristics | ||
| Disease history, N (%) | ||
| De Novo AML | 38 (49) | 18 (47) |
| Secondary AML | 40 (51) | 20 (53) |
| Prior hypomethylating agent (decitabine or azacitidine) use, N (%) | 11 (14) | 6 (16) |
| ECOG PSα, N (%) | ||
| 0 to 1 | 36 (46) | 20 (53) |
| 2 | 41 (53) | 18 (47) |
| Cytogenetic risk status, N (%) | ||
| Good/Intermediate | 49 (63) | 21 (55) |
| Poor | 29 (37) | 17 (45) |
| Baseline severe cardiac disease, N (%) | 52 (67) | 20 (53) |
| Baseline serum creatinine > 1.3 mg/dL, N (%) | 15 (19) | 5 (13) |
Abbreviations: AML=acute myeloid leukaemia; ECOG PS=Eastern Cooperative Oncology Group Performance Status;
N=number of patients.
a Baseline ECOG PS was not reported for one patient in the Daurismo with low-dose cytarabine arm.
Table 8. AML efficacy results from Study 1:
| Endpoint/study population | Daurismo with low-dose cytarabine | Low-dose cytarabine alone |
|---|---|---|
| OS in AML study population | N=78 | N=38 |
| Median survival, months (95% CI) | 8,3 (4,7, 12,2) | 4,3 (1,9, 5,7) |
| Hazard ratio (95% CI)a | 0,463 (0,299, 0,717) | |
| p-valueb | 0,0002 | |
| OS in de novo AML study population | N=38 | N=18 |
| Median survival, months (95% CI) | 6,6 (3,7, 12,4) | 4,3 (1,3, 10,7) |
| Hazard ratio (95% CI)a | 0,670 (0,362, 1,239) | |
| p-valueb | 0,0991 | |
| OS in secondary AML study population | N=40 | N=20 |
| Median survival, months (95% CI) | 9,1 (4,4, 16,5) | 4,1 (1,5, 6,4) |
| Hazard ratio (95% CI)a | 0,287 (0,151, 0,548) | |
| p-valueb | <0,0001 | |
| Good/intermediate cytogenetic risk group | N=49 | N=21 |
| Median survival, months (95% CI) | 11,1 (7,1, 14,9) | 4,4 (1,8, 8,7) |
| Hazard ratio (95% CI)a | 0,417 (0,233, 0,744) | |
| p-valueb | 0,0011 | |
| Poor cytogenetic risk group | N=29 | N=17 |
| Median survival, months (95% CI) | 4,4 (3,4, 9,1) | 3,1 (1,1, 6,4) |
| Hazard ratio (95% CI)a | 0,528 (0,273, 1,022) | |
| p-valueb | 0,0269 | |
Abbreviations: AML=acute myeloid leukaemia; CI=confidence interval; N=number of patients; OS=overall survival.
a Hazard ratio (Daurismo with low-dose cytarabine/low-dose cytarabine alone) based on the Cox Proportional hazards model stratified by prognosis stratum.
b 1-sided p-value from stratified log-rank test based on cytogenetic risk.
Figure 1. Kaplan-Meier plot of overall survival for AML patients:
Abbreviations: CI=confidence interval; LDAC=low-dose cytarabine; OS=overall survival.
Improvement in OS was consistent across pre-specified subgroups by cytogenetic risk.
Based on investigator reported response, a numerically higher complete response (CR) rate (defined as absolute neutrophil count ≥1000/μl, platelet count ≥100,000/μl, <5% bone marrow blasts, transfusion independent, and no extramedullary disease) was achieved for AML patients in the Daurismo with low-dose cytarabine arm (17.9% [95% CI: 9.4%, 26.5%]) vs the low-dose cytarabine alone arm (2.6% [95% CI: 0.0%, 7.7%]).
Paediatric population
The European Medicines Agency has waived the obligation to submit the results of studies with Daurismo in all subsets of the paediatric population in treatment of AML (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Absorption
Following a single 100 mg dose of glasdegib, peak concentration in plasma israpidly reached with the median Tmax of 2 hours. Following repeat 100 mg once daily dosing to steady state, glasdegib median Tmax ranged from approximately 1.3 hours to 1.8 hours.
Food effect
After oral administration of glasdegib tablets, the mean absolute bioavailability is 77.1% compared to intravenous administration. Administration of glasdegib with a high-fat, high-calorie meal resulted in 16% lower exposure (AUCinf) compared to overnight fasting. The impact of food on the pharmacokinetics of glasdegib is not considered clinically relevant. Glasdegib may be administered with or without food.
Following 100 mg once daily glasdegib dosing, the mean (coefficient of variation, CV) of glasdegib Cmax was 1,252 ng/mL (44) and AUCtau was 17,210 ng•hr/mL (54%) in patients with cancer.
Distribution
Glasdegib is 91% bound to human plasma proteins in vitro. The mean (%CV) apparent volume of distribution (Vz/F) was 188 (20) L following a single dose of 100 mg glasdegib in patients with haematologic malignancies.
Biotransformation
The primary metabolic pathways for glasdegib were comprised of N-demethylation, glucuronidation, oxidation, and dehydrogenation. In plasma, the N-desmethyl and N-glucuronide metabolites of glasdegib accounted for 7.9% and 7.2% of the circulating radioactivity, respectively. Other metabolites in plasma individually accounted for <5% of circulating radioactivity.
In vitro interaction studies
In vitro CYP inhibition and induction: In vitro studies indicated that glasdegib is not an inhibitor of CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, and CYP3A4/5 at clinically relevant concentrations. In vitro studies indicated that glasdegib is not an inducer of CYP1A2, CYP2B6, or CYP3A4 at clinically relevant concentrations.
In vitro studies of UGT inhibition: In vitro studies indicated that glasdegib is not an inhibitor of uridine-diphosphate glucuronosyltransferase (UGT)1A4, UGT1A6, UGT2B7, and UGT2B15 at clinically relevant concentrations. Glasdegib may have the potential to inhibit UGT1A1, and possibly UGT1A9, however, clinically relevant drug-drug interactions are not expected.
In vitro studies of organic anion and cation transporter inhibition: In vitro studies indicated that glasdegib is not an inhibitor of organic anion transporting polypeptide (OATP)1B1, OATP1B3, organic anion transporter (OAT)1, OAT3, organic cation transporter (OCT)2, at clinically relevant concentrations.
Elimination
The mean (± SD) plasma half-life of glasdegib was 17.4 ± 3.7 hours after a single dose of 100 mg glasdegib in patients. The geometric mean oral clearance after multiple dosing was 6.45 L/hr. Following oral administration of a 100 mg radiolabeled dose of glasdegib to healthy subjects, mean 48.9% and 41.7% of the radioactivity dosed was recovered in urine and faeces, respectively. The overall mean mass balance of the dosed radioactivity in the excreta was 90.6%. Unchanged glasdegib was the major component of human plasma, accounting for 69.4% of the total drug-related material. Unchanged glasdegib recovered in the urine and faeces accounted for 17.2% and 19.5% of the dose, respectively.
Linearity / non-linearity
The steady state systemic glasdegib exposure (Cmax and AUCtau) increased in a dose-proportional manner over the dosing range of 5 mg to 600 mg once daily.
Special populations
Hepatic impairment
Data from a dedicated pharmacokinetic trial have shown that plasma exposures for total glasdegib (AUCinf and Cmax) were similar between subjects with normal hepatic function and subjects with moderate hepatic impairment (Child-Pugh Class B), whilst geometric mean AUCinf and Cmax values were 24% and 42% lower, respectively, for subjects with severe hepatic impairment (Child-Pugh Class C), compared to the normal hepatic function group. The glasdegib unbound exposure (unbound AUCinf) is increased by 18% and 16% in subjects with moderate and severe impairment, respectively, relative to subjects with normal hepatic function. Peak glasdegib unbound exposure (unbound Cmax) increased by 1%, for moderate hepatic impairment and decreased by 11% for severe hepatic impairment, relative to subjects with normal hepatic function. These changes are not considered to be clinically relevant.
Renal impairment
Data from a dedicated pharmacokinetic trial in subjects with varying degrees of renal function impairment indicate that total glasdegib exposure (AUCinf) increased by 105%, and 102 % with moderate (30 mL/min ≤ eGFR <60 mL/min), and severe (eGFR <30 mL/min) renal impairment, respectively, relative to subjects with normal (eGFR ≥90 mL/min) renal function. Peak glasdegib exposure (Cmax) increased by 37%, and 20% for subjects with moderate, and severe renal impairment, respectively, relative to subjects with normal renal function. These changes are not considered to be clinically relevant.
Elderly
In patients assigned to treatment with Daurismo with low-dose cytarabine (n=88; Study 1), 97.7% of the patients were aged 65 or older and 60.2% of the patients were aged 75 or older. Study 1 did not include a sufficient number of patients younger than age 65 to determine differences in adverse reactions reported from patients older than 65.
Age, race, gender, and body weight
There are limited data in patients younger than 65 years of age. Population pharmacokinetic analyses in adult patients (n=269) indicate that there are no clinically relevant effects of age, gender, race, body weight on the pharmacokinetics of glasdegib.
5.3. Preclinical safety data
The primary target organ findings following repeat oral administration of glasdegib in rats and dogs for up to 26 and 39 weeks in duration, respectively, included the kidney (degeneration/necrosis) in rat and dog, the liver (necrosis/inflammation) in dog only, and the testis (degeneration), growing incisor teeth (necrosis/broken), growing bone (partial to full closure of epiphysis), and peripheral nerve (axonal degeneration) in rat only. Additional clinical observations of alopecia, weight loss, and muscle tremors/twitching, known class effects of SMO inhibitors, were observed in both species. These systemic toxicities were generally dose-dependent and observed at exposures ranging from approximately <0.03 to 8-times the clinically relevant exposure based on nonclinical to clinical comparison of the observed unbound AUC at the recommended clinical dose of 100 mg once daily.
Complete reversibility of toxicities to the kidney (degeneration/necrosis), peripheral nerve (axonal degeneration), seminiferous tubule (testicular degeneration), and the clinical observations of muscle tremors/twitching was demonstrated following up to 16-week recovery, whereas partial recovery was demonstrated in the liver (necrosis/inflammation). The observation of alopecia, bone and teeth effects, and testicular hypospermatogenesis did not recover. In addition, QTc prolongation was identified in telemetered dogs at unbound Cmax exposures approximately 4-times the observed unbound Cmax exposure at the recommended clinical dose of 100 mg once daily.
Glasdegib was not mutagenic in vitro in the bacterial reverse mutation (Ames) assay and was not clastogenic in the in vitro chromosome aberration assay in human lymphocytes. Glasdegib was not clastogenic or aneugenic in the rat micronucleus assay.
Carcinogenicity studies have not been conducted with glasdegib.
In repeat-dose toxicity studies in rats, findings observed in the male reproductive tract included adverse testicular changes with glasdegib at doses 50 mg/kg/day, and consisted of minimal to severe hypospermatogenesis characterised by partial to complete loss of spermatogonia, spermatocytes and spermatids and testicular degeneration. Hypospermatogenesis did not recover whereas testicular degeneration did recover. The dose at which adverse testicular effects were observed in male rats was identified as 50 mg/kg/day with corresponding systemic exposures that were approximately 8-times those associated with the observed human exposure at the 100 mg once daily dose (based on unbound AUC in respective species). Safety margin for NOAEL (10 mg/kg/day) is 0.6, hence lower than clinically relevant.
In embryo-foetal developmental toxicity studies conducted in rats and rabbits, glasdegib was severely toxic to the conceptus as evidenced by complete resorption and/or abortion of foetuses, and teratogenic effects at lower dose levels. Teratogenic effects included craniofacial malformations, malformed limbs, paws/digits, trunk and tail, dilation of brain, malpositioned/malformed eyes, misshapen head, small tongue, absent palate, teeth and viscera, diaphragmatic hernia, oedema, persistent truncus arteriosus, heart defects, absent lung, absent trachea, rib and vertebral abnormalities, and malformed or absent structures in the appendicular skeleton (notably the long bones). Severe developmental malformations were observed at maternal systemic exposures lower than the relevant human exposure at the recommended dose of 100 mg once daily.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.