DIPHERELINE Powder and solvent for suspension for injection Ref.[50753] Active ingredients: Triptorelin
Source: Pharmaceutical Benefits Scheme (AU) Revision Year: 2022 Publisher: Ipsen Pty Ltd, Level 2, Building 4, Brandon Office Park, 540 Springvale Road, Glen Waverley Victoria 3150 Telephone: 1800 317 033
Product name and form
Triptorelin embonate.
| Pharmaceutical Form |
|---|
|
Powder and solvent for suspension for injection, prolonged release granules. White to off-white powder. |
Qualitative and quantitative composition
Diphereline 3.75mg 1 month formulation: Each vial contains a triptorelin content which allows the administration of an effective dose of 3.75 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 1.875 mg of triptorelin.
Diphereline 11.25mg 3 month formulation: Each vial contains a triptorelin content which allows the administration of an effective dose of 11.25 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 5.625 mg of triptorelin.
Diphereline 22.5mg 6 month formulation: Each vial contains a triptorelin content which allows the administration of an effective dose of 22.5 mg triptorelin. After reconstitution in 2 mL of solvent, 1 mL of reconstituted suspension contains 11.25 mg of triptorelin.
For the full list of excipients, see Section 6.1 LIST OF EXCIPIENTS.
Physicochemical properties
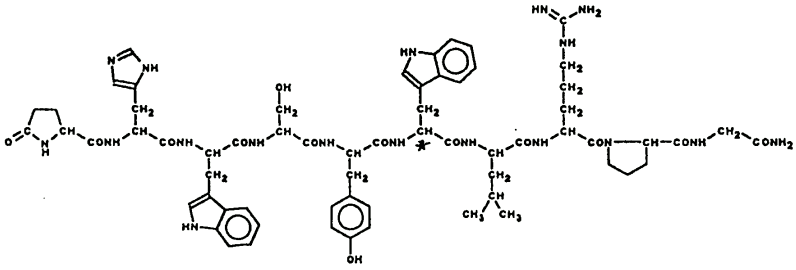
Chemical structure:
[(L-Pyr)-(L-His)-(L-Trp)-(L-Ser)-(L-Tyr)-(D-Trp)-(L-Leu)-(L-Arg)-(L-Pro)-(Gly-NH2)]
Molecular formula: C64H82N18O13 (triptorelin). C23H16O6 (embonate)
Molecular weight: 1311.5 (triptorelin) + 388.4 (embonate)
CAS number: 57773-63-4
| Active Ingredient | Description | |
|---|---|---|
| Triptorelin |
Triptorelin is a decapeptide analogue of GnRH which initially stimulates release of pituitary gonadotropins. |
| List of Excipients |
|---|
|
Composition of the Powder: Polyglactin Composition of the Solvent: Water for Injections |
Pack sizes and marketing
Diphereline 3.75 mg 1 month formulation: 6 mL type I brown tint glass vial with grey bromobutyl stopper and purple aluminium flip-off capsule.
Diphereline 11.25 mg 3 month formulation: 6 mL type I brown tint glass vial with grey bromobutyl stopper and yellow green aluminium flip-off capsule.
Diphereline 22.5 mg 6 month formulation: 6 mL type I brown tint glass vial with grey bromobutyl stopper and dark green aluminium flip-off capsule.
Solvent: Type I glass ampoule containing 2 mL of sterile solvent for suspension.
Each box contains 1 vial, 1 ampoule and 1 blister containing 1 empty polypropylene injection syringe and 2 injection needles.
Marketing authorization holder
Ipsen Pty Ltd, Level 2, Building 4, Brandon Office Park, 540 Springvale Road, Glen Waverley Victoria 3150
Telephone: 1800 317 033
Marketing authorization dates and numbers
Date of first approval: 28 August 2006
Drugs
| Drug | Countries | |
|---|---|---|
| DIPHERELINE | Australia, Ecuador, Estonia, Hong Kong, Lithuania, Poland, Romania, Singapore |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.