EMOCLOT Powder and solvent for solution for infusion Ref.[51154] Active ingredients: Coagulation factor VIII
Source: Medicines Authority (MT) Revision Year: 2020 Publisher: Kedrion S.p.A. Loc. Ai Conti, 55051 Castelvecchio Pascoli, Barga (Lucca)
4.1. Therapeutic indications
Treatment and prophylaxis of bleeding in patients with haemophilia A (congenital Factor VIII deficiency).
Management of acquired Factor VIII deficiency.
Treatment of haemophiliacs with antibodies against Factor VIII (inhibitors: see also 4.4).
4.2. Posology and method of administration
Treatment should be initiated under the supervision of a physician experienced in the treatment of haemophilia.
Treatment monitoring
During the course of treatment, appropriate determination of factor VIII levels is advised to guide the dose to be administered and the frequency of repeated infusions. Individual patients may vary in their response to factor VIII, demonstrating different half-lives and recoveries. Dose based on bodyweight may require adjustment in underweight or overweight patients.
In the case of major surgical interventions in particular, precise monitoring of the substitution therapy by means of coagulation analysis (plasma factor VIII activity) is indispensable.
When using an in vitro thromboplastin time (aPTT)-based one stage clotting assay for determining factor VIII activity in patients' blood samples, plasma factor VIII activity results can be significantly affected by both the type of aPTT reagent and the reference standard used in the assay. Also there can be significant discrepancies between assay results obtained by aPTT-based one stage clotting assay and the chromogenic assay according to Ph. Eur. This is of importance particularly when changing the laboratory and/or reagents used in the assay.
Posology
The dosage and duration of the substitution therapy depend on the severity of the factor VIII deficiency, on the location and extent of the bleeding and on the patient’s clinical condition.
The number of units of factor VIII administered is expressed in International Units (IU), which are related to the current WHO standard for factor VIII products. Factor VIII activity in plasma is expressed either as a percentage (relative to normal human plasma) or in International Units (relative to an international standard for factor VIII in plasma). One International Unit (IU) of factor VIII activity is equivalent to that quantity of factor VIII in one ml of normal human plasma.
On demand treatment
The calculation of the required dose of factor VIII is based on the empirical finding that 1 International Unit (IU) of factor VIII per kg of body weight raises the plasma factor VIII activity by 1.5% to 2% of normal activity.
The required dosage is determined using the following formula:
Required units = body weight (kg) x desired factor VIII rise (%) (IU/dl) x 0.4
The amount to be administered and the frequency of administration should always be oriented to the clinical effectiveness in the individual case.
In the case of the following haemorrhagic events, the factor VIII activity should not fall below the given plasma activity level (in % of normal) in the corresponding period. The following table can be used to guide dosing in bleeding episodes and surgery:
| Degree of haemorrhage / Type of surgical procedure | Factor VIII level required (%) (IU/dl) | Frequency of doses (hours)/Duration of therapy (days) |
|---|---|---|
| Haemorrhage Early haemarthrosis, muscle bleeding or oral bleeding More extensive haemarthrosis, muscle bleeding or haematoma Life-threatening haemorrhages | 20–40 30–60 60–100 | Repeat every 12 to 24 hours. At least 1 day, until the bleeding episode as indicated by pain is resolved or healing is achieved. Repeat infusion every 12-24 hours for 3–4 days or more until pain and acute disability are resolved. Repeat infusion every 8 to 24 hours until threat is resolved. |
| Surgery Minor surgery including tooth extraction | 30–60 | Every 24 hours, at least 1 day, until healing is achieved. |
| Major surgery | 80–100 (pre- and post- operative) | Repeat infusion every 8-24 hours until adequate wound healing, then therapy for at least another 7 days to maintain a factor VIII activity of 30% to 60% (30 IU/dl – 60 IU/dl). |
Prophylaxis
For long term prophylaxis against bleeding in patients with severe haemophilia A, the usual doses are 20 to 40 IU of factor VIII per kg body weight at intervals of 2 to 3 days. In some cases, especially in younger patients, shorter dosage intervals or higher doses may be necessary.
Paediatric population
The safety and efficacy of EMOCLOT in children under 12 years have not yet been established. Currently available data are described in section 5.1 but posology recommendations cannot be made. The posology in adolescents (12-18 years) for each indication is calculated on body weight.
Method of administration
Intravenous use, by injection or slow infusion.
In the case of intravenous injection, it is recommended to observe an administration time of 3 to 5 minutes, checking the pulse rate of the patients and interrupting administration or decreasing the speed of injection if pulse rate rises.
The infusion rate should be evaluated for each patient.
For instructions on reconstitution of the medicinal product before administration, see section 6.6.
4.9. Overdose
No symptoms of overdose with human plasma coagulation factor VIII have been reported.
6.3. Shelf life
3 years.
Reconstituted product should be used immediately.
6.4. Special precautions for storage
Store in a refrigerator (2°C-8°C). Do not freeze.
Keep the vial in the outer carton, in order to protect from light.
Before use and within the shelf-life, the powder vial can be stored at room temperature, not exceeding 25°C, for a maximum of 6 consecutive months.
After this period, the powder vial must be discarded. In any case, this vial can no longer be put back in the refrigerator if kept at room temperature.
The starting date of the storage at room temperature should be reported on the outer box.
6.5. Nature and contents of container
One neutral type I glass vial with type I elastomer stopper containing the powder; one neutral type I glass vial with type I elastomer stopper containing the solvent; a non pyrogenic, sterile, disposable set consisting of a medical device for reconstitution, a syringe for injection and a butterfly needle with PVC tube.
EMOCLOT 1000 IU/10 ml Powder and solvent for solution for infusion. One vial of powder + one vial of solvent + set for reconstitution and administration.
6.6. Special precautions for disposal and other handling
Reconstitution of the powder with the solvent:
1. bring the vial of powder and the vial of solvent to room temperature;
2. this temperature must be maintained during the entire reconstitution process (maximum 10 minutes);
3. remove the protection caps of the powder and solvent vials;
4. clean the surfaces of the stoppers of the two vials with alcohol;
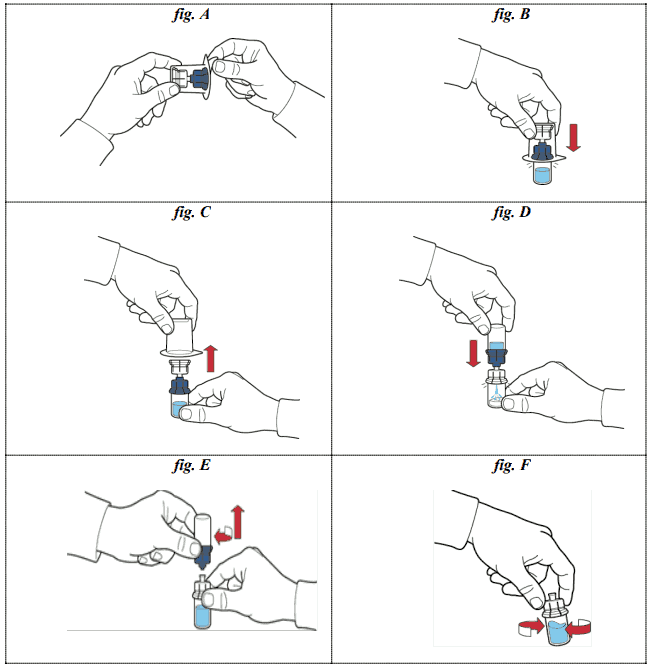
5. open the package of the device by peeling away the upper lid; attention should be taken not to touch the internal part (fig. A);
6. do not remove the device from the package;
7. turn the box of the device upside down and insert the plastic spike through the solvent vial stopper so that the blue part of the device is connected to the solvent vial (fig. B);
8. hold the edge of the device box and slip out the device without touching it (fig. C);
9. make sure that the powder vial is placed on a secure surface, turn the system upside down so that the solvent vial is on top of the device; press the transparent adapter on the powder vial stopper so that the plastic spike passes through the powder vial stopper; the solvent will be aspired automatically into the powder vial (fig. D);
10. after the transfer of the solvent unscrew the blue part of the transfer system, to which the solvent vial is attached, and remove it (fig. E);
11. shake gently the vial until complete dissolution of the powder (fig. F);
12. do not shake vigorously, foaming should be avoided.
Administration of the solution:
After reconstitution, the solution may contain a few small filaments or particles.
Reconstituted product should be inspected visually for particulate and discoloration prior to administration. The solution should be clear or slightly opalescent. Do not use solutions that are cloudy or have deposits.
1. Fill the syringe with air, pulling back the plunger, connect it to the device and inject air into the powder vial containing the reconstituted solution (fig. G);
2. keeping the plunger still, turn the system upside down so that the powder vial containing the reconstituted solution is on top of the device and aspire the concentrate into the syringe by slowly pulling the plunger back (fig. H);
3. disconnect the syringe by turning it anti-clockwise;
4. visually inspect the solution in the syringe, it should be clear or slightly opalescent, without particles;
5. connect the butterfly needle to the syringe and infuse or slowly inject intravenously.
Once the vials have been opened, the content must be used immediately.
The reconstituted solution transferred into the syringe should be used immediately.
The content of the vial should be used in a single administration.
Do not use after the expiry date stated on the label.
Any unused product or waste material should be disposed of in accordance with local requirements.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.