GOMEKLI Capsule / Tablet Ref.[114757] Active ingredients: Mirdametinib
Source: FDA, National Drug Code (US) Revision Year: 2025
Product description
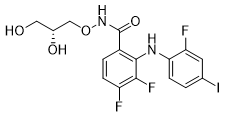
GOMEKLI capsules and tablets for oral suspension contain mirdametinib, a kinase inhibitor. Mirdametinib is chemically known as (R)N(2,3-dihydroxypropoxy)-3,4-difluoro-2-((2-fluoro-4-iodophenyl)amino) benzamide. The molecular formula is C16H14F3IN2O4 and the molecular weight is 482.20 g/mol. The structural formula for mirdametinib is:
Mirdametinib is a white to tan or pink solid with an aqueous solubility of 0.25 mg/mL and a pH of 7.2 in water at 25°C. The molecule has a pKa of 7.96.
GOMEKLI capsules and tablets for oral suspension are immediate release (IR) dosage forms intended for oral administration.
GOMEKLI (mirdametinib) 1 mg and 2 mg capsules contain 1 mg and 2 mg mirdametinib, respectively, in gelatin capsule and the following inactive ingredients: croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The gelatin capsule shell contains FD&C blue #1, gelatin, titanium dioxide, and yellow iron oxide. The capsule is imprinted with white ink that contains butyl alcohol, dehydrated alcohol, isopropyl alcohol, potassium hydroxide, propylene glycol, purified water, shellac, strong ammonia solution, and titanium dioxide.
GOMEKLI (mirdametinib) 1 mg tablets for oral suspension contain 1 mg mirdametinib and the following inactive ingredients: croscarmellose sodium, magnesium stearate, microcrystalline cellulose, grape flavor, and sucralose. The grape flavor includes corn syrup solids, modified corn starch, and triacetin.
| Dosage Forms and Strengths |
|---|
|
Capsules:
Tablets for Oral Suspension:
|
| How Supplied | ||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
|
GOMEKLI Capsules are supplied as follows:
GOMEKLI Tablets for Oral Suspension are supplied as follows:
Manufactured for: SpringWorks Therapeutics, Inc., Stamford, CT 06902 |
Drugs
| Drug | Countries | |
|---|---|---|
| GOMEKLI | United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.