KAPRUVIA Solution for injection Ref.[49880] Active ingredients: Difelikefalin
Source: European Medicines Agency (EU) Revision Year: 2022 Publisher: Vifor Fresenius Medical Care Renal Pharma France, 100–101 Terrasse Boieldieu, Tour Franklin La Défense 8, 92042 Paris la Défense Cedex, France
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: all other therapeutic products, other therapeutic products
ATC code: V03AX04
Mechanism of action
Difelikefalin is a selective kappa opioid receptor agonist with low central nervous system penetration. The physicochemical properties of difelikefalin (hydrophilic, synthetic D-amino acid peptide with high polar surface area and charge at physiological pH) minimize its passive diffusion (permeability) and active transport across membranes, thus limiting penetration into the central nervous system.
The pathophysiology of chronic kidney disease-associated pruritus is thought to be multifactorial, including systemic inflammation and an imbalance in the endogenous opioid system (e.g., overexpression of mu opioid receptors and concomitant downregulation of kappa opioid receptors). Opioid receptors are known to modulate itch signals and inflammation, with kappa opioid receptor activation reducing itch and producing immunomodulatory effects.
The activation of kappa opioid receptors on peripheral sensory neurons and immune cells by difelikefalin are considered mechanistically responsible for the antipruritic and anti-inflammatory effects.
Clinical efficacy and safety
Placebo-controlled studies
In two pivotal clinical phase-3 studies of similar double-blind, randomised, placebo-controlled design (KALM-1 and KALM-2), chronic kidney disease patients on haemodialysis with moderate-to-severe pruritus received either placebo or 0.5 micrograms/kg difelikefalin intravenously 3 times a week following haemodialysis for 12 weeks. A maximum of 4 doses per week was allowed in patients receiving an additional dialysis during a given week. The primary endpoint in both studies was the percentage of patients who achieved at least a 3-point reduction from baseline in the Worst Itching-Numerical Rating Scale (WI-NRS) at 12 weeks. The main secondary endpoints in both studies were the percentages of patients with an improvement in the WI-NRS of at least 4 points after 12 weeks and the changes in itch severity and itch-related quality of life (QoL) as measured by the total Skindex-10 and the 5-D Itch scale. A responder analysis based on Patient Global Impression of Change was also included.
A total of 851 patients with moderate-to-severe pruritus (baseline WI-NRS >4) were enrolled in the pivotal studies. Mean age was 59 years, 33.1% were aged 65 and over and 11.1% were aged 75 and over; 60% of patients were male. The baseline mean WI-NRS scores were 7.18 in both, difelikefalin and placebo arms; baseline median WI-NRS scores were 7.13 (range 4.2 to 10) in difelikefalin and 7.13 (range 4.1 to 10) in placebo arm. Other disease characteristics at baseline were comparable in difelikefalin and placebo arms: time from diagnosis of chronic kidney disease (8.22 years vs. 8.54 years), duration of pruritus (3.20 years vs. 3.31 years) and use of medicinal products intended to relieve pruritus such as antihistamines, corticosteroids, gabapentin or pregabalin (37.5% vs. 38%). Across studies, difelikefalin significantly reduced itch intensity and improved itch-related QoL over 12 weeks as shown in Table 2.
Table 2. Summary of primary and key secondary outcomes in KALM-1 and KALM-2 at week 12:
| Endpoint by end of week 12 | KALM-1 (n=378) | KALM-2 (n=473) | ||
|---|---|---|---|---|
| difelikefalin (n=189) | Placebo (n=189) | difelikefalin (n=237) | Placebo (n=236) | |
| Primary endpoint | ||||
| WI-NRS | ||||
| Patients with ≥ 3-point improvement (%) | 51.0% (p<0.001) | 27.6% | 54.0% (p=0.02) | 42.2% |
| WI-NRS | ||||
| Patients with ≥ 4-point improvement (%) | 38.9% (p<0.001) | 18.0% | 41.2% (p=0.01) | 28.4% |
| Skindex-10 | ||||
| Change from baseline [total score] | -17.2 (p<0.001) | -12.0 | -16.6 (p=0.171) | -14.8 |
| 5-D Itch | ||||
| Change from baseline [total score] | -5.0 (p<0.001) | -3.7 | -4.9 Not applicable1 | -3.8 |
1 Was not tested based on the hierarchical testing order.
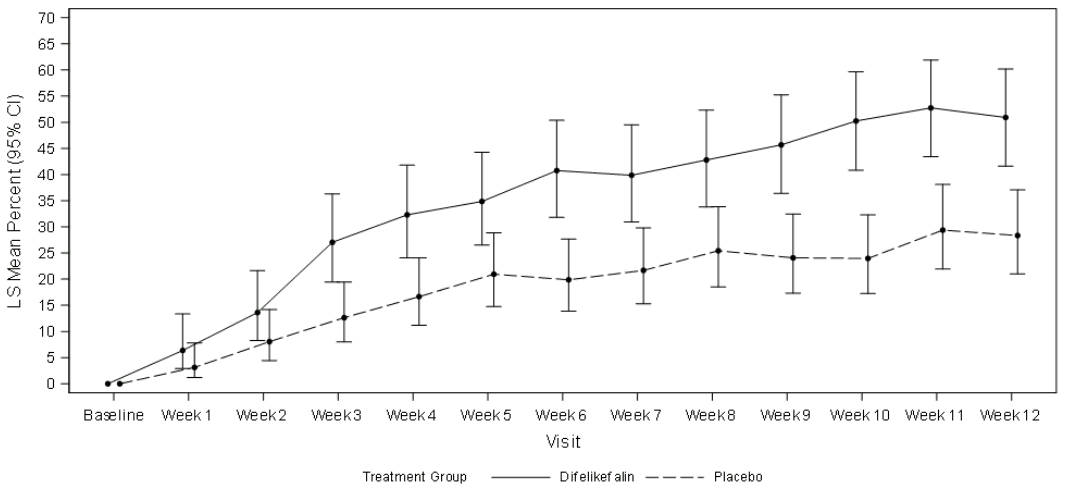
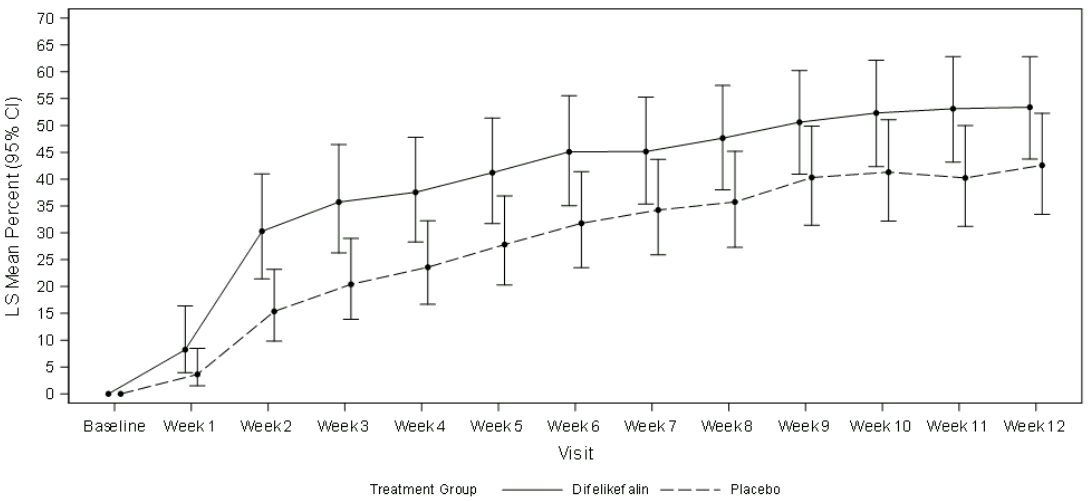
Figure 1 shows the mean percentage from KALM-1 and KALM-2 with a ≥ 3-point improvement from baseline in WI-NRS score by study week. Based on odds ratios, statistically significant improvements favouring the difelikefalin group were seen by week 3 in KALM-1 and by week 2 in KALM-2 and continued at each subsequent week through week 12 in both studies.
Figure 1. Percentage of patients with ≥ 3-point improvement with respect to WI-NRS score by week in KALM-1 and KALM-2 (ITT population) KALM-1:
KALM-1:
CI = confidence interval; ITT = intent to treat; LS = least squares; WI-NRS = Worst Itching-Numerical Rating Scale
KALM-2:
CI = confidence interval; ITT = intent to treat; LS = least squares; WI-NRS = Worst Itching-Numerical Rating Scale
Open label extension studies
The effect of treatment with difelikefalin for up to 52 weeks was evaluated using the 5-D Itch Scale in single arm, open label extensions of studies KALM-1 and KALM-2 including 712 patients. In patients switching from placebo to difelikefalin at the end of the double-blind phase, an improvement in 5-D Itch score was observed after 4 weeks of treatment, with an LS mean (SE) of the change from baseline comparable to the patients receiving difelikefalin from study start: -6.0 (0.22) vs. -5.7 (0.23). The improvement in 5-D Itch score was maintained in both treatment groups throughout the 52-week treatment.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with difelikefalin in one or more subsets of the paediatric population in the treatment of chronic kidney disease associated pruritus (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
In patients with severe renal impairment undergoing haemodialysis, total body clearance of difelikefalin is reduced compared to healthy subjects and plasma concentrations decrease slowly until cleared during dialysis. Due to the 70-80% of difelikefalin removed during dialysis, difelikefalin is administered after each haemodialysis session in these patients. The available data on interindividual variability in haemodialysis subjects receiving 0.5 microgram/kg difelikefalin suggest that variability of AUC can exceed 30%.
Distribution
Plasma protein binding of difelikefalin is low to moderate, ranging from 24-32%, and remains unaffected by renal impairment. Mean volume of distribution at steady state ranged from 145 to 189 mL/kg in healthy subjects and from 214 to 301 mL/kg in haemodialysis patients with moderate-to-severe pruritus. Difelikefalin penetration into the central nervous system is limited (below limit of quantification) as shown by physico-chemical, in-vitro and animal data.
Elimination
In healthy subjects, the primary route of elimination for difelikefalin is renal, accounting for about 81% of the dose being excreted in urine as compared to 11% via faecal excretion. In both healthy volunteers and subjects on haemodialysis, most of the dose excreted into urine and faeces was unchanged difelikefalin with minor quantities of putative metabolites, none exceeding 2.5%. Mean total clearance ranged from 54 to 71 mL/h/kg and mean half-lives from 2 to 3 hours. By contrast, in renally impaired haemodialysis patients, elimination was predominantly via faeces, accounting on average for about 59% of the dose; about 19% were recovered in dialysate and about 11% were found in urine. As compared to subjects with normal renal function, mean total clearance decreased and half-lives increased about 10-fold with ranges of 5.3 to 7.5 mL/h/kg and 23 to 31 hours, respectively.
Interaction with other medicinal products
Difelikefalin is neither a substrate for CYP1A2, CYP2C8, CYP2C9, CYP2C19, CYP2D6, or CYP3A4, nor an inhibitor of CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6, or CYP3A4/5 and has minimal to no potential for induction of human CYP1A2, CYP2B6, or CYP3A. It is not an inhibitor of glucuronidation enzymes either (UGT1A3, UGT1A9, or UGT2B7).
In addition, difelikefalin is not an inhibitor of BCRP, BSEP, LAT1, MATE1, MATE2-K, MRP2, OAT1, OAT3, OATP1A2, OATP1B1, OATP1B3, OCT1, OCT2, OCT3, P-glycoprotein, PEPT1 or PEPT2, and is not a substrate for ASBT, BCRP, BSEP, LAT1, MATE1, MATE2-K, MRP2, OAT1, OAT2, OAT3, OATP1A2, OATP1B1, OATP1B3, OATP2B1, OCT1, OCT2, OCT3, OCTN1, OCTN2, OSTαβ, P-glycoprotein, PEPT1 or PEPT2.
Linearity/non-linearity
Pharmacokinetics of difelikefalin were demonstrated to be linear and dose-proportional in healthy subjects (tested over dose ranges of 1 to 40 and 1 to 20 micrograms/kg in single and repeated dose studies, respectively). Steady state dose proportionality was also established in chronic kidney disease patients on haemodialysis receiving repeated doses from 0.5 to 2.5 micrograms/kg, 3 times per week for 1 week. However, in another study dose proportionality was observed at doses of 0.5 and 1 micrograms/kg, but not at the dose of 1.5 micrograms/kg. Trough plasma concentration values reached steady state by the second dose and for the dose of 0.5 micrograms/kg, mean accumulation ratio was 1.144 in one study based on AUC0-48h and 1.33 in another study, based on AUC0-44h; showing that variability for accumulation parameters can exceed 30%.
Characteristics in specific groups of subjects or patients
Based on available evidence, there is no indication that factors such as age, sex, ethnicity, or mild to moderate hepatic impairment have any impact on the pharmacokinetics of difelikefalin.
5.3. Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeat-dose toxicity, genotoxicity and carcinogenic potential.
Reproductive toxicity
In rats, male and female fertility, early embryonic, and prenatal and postnatal development were not affected up to 2000-fold the human AUC. In the rabbit, prenatal development was neither impaired despite marked maternal toxicity at 30-fold the human AUC.
Difelikefalin crosses the placenta in rats.
Abuse and dependence potential
The abuse and dependence potential studies in the rat suggest that difelikefalin is not likely to present a risk of physical dependence or abuse potential.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.