OCREVUS Concentrate for solution for infusion Ref.[8966] Active ingredients: Ocrelizumab
Source: European Medicines Agency (EU) Revision Year: 2020 Publisher: Roche Registration GmbH, Emil-Barell-Strasse 1, 79639, Grenzach-Wyhlen, Germany
Pharmacodynamic properties
Pharmacotherapeutic group: selective immunosuppressants group
ATC code: L04AA36
Mechanism of Action
Ocrelizumab is a recombinant humanised monoclonal antibody that selectively targets CD20-expressing B cells.
CD20 is a cell surface antigen found on pre-B cells, mature and memory B cells but not expressed on lymphoid stem cells and plasma cells.
The precise mechanisms through which ocrelizumab exerts its therapeutic clinical effects in MS is not fully elucidated but is presumed to involve immunomodulation through the reduction in the number and function of CD20-expressing B cells. Following cell surface binding, ocrelizumab selectively depletes CD20-expressing B cells through antibody-dependent cellular phagocytosis (ADCP), antibody-dependent cellular cytotoxicity (ADCC), complement-dependent cytotoxicity (CDC), and apoptosis. The capacity of B-cell reconstitution and preexisting humoral immunity are preserved. In addition, innate immunity and total T-cell numbers are not affected.
Pharmacodynamic effects
Treatment with Ocrevus leads to rapid depletion of CD19+ B cells in blood by 14 days post treatment (first time-point of assessment) as an expected pharmacologic effect. This was sustained throughout the treatment period. For the B cell counts, CD19 is used, as the presence of Ocrevus interferes with
the recognition of CD20 by the assay. In the Phase III studies, between each dose of Ocrevus, up to 5% of patients showed B-cell repletion (> lower limit of normal (LLN) or baseline) at least at one time point. The extent and duration of B-cell depletion was consistent in the PPMS and RMS trials.
The longest follow up time after the last Ocrevus infusion (Phase II study WA21493, N=51) indicates that the median time to B-cell repletion (return to baseline/LLN whichever occurred first) was 72 weeks (range 27-175 weeks). 90% of all patients had their B-cells repleted to LLN or baseline by approximately two and a half years after the last infusion.
Clinical efficacy and safety
Relapsing forms of MS
Efficacy and safety of Ocrevus were evaluated in two randomised, double-blind, double-dummy, active comparator-controlled clinical trials (WA21092 and WA21093), with identical design, in patients with relapsing forms of MS (in accordance with McDonald criteria 2010) and evidence of disease activity (as defined by clinical or imaging features) within the previous two years. Study design and baseline characteristics of the study population are summarised in Table 3.
Demographic and baseline characteristics were well balanced across the two treatment groups. Patients receiving Ocrevus (Group A) were given 600 mg every 6 months (Dose 1 as 2 x 300 mg intravenous infusions, administered 2 weeks apart, and subsequent doses were administered as a single 600 mg intravenous infusion). Patients in Group B were administered Interferon beta-1a (Rebif) 44 mcg via subcutaneous injection 3 times per week.
Table 3. Study Design, Demographic and Baseline Characteristics:
| Study 1 | Study 2 | |||
|---|---|---|---|---|
| Study name | WA21092 (OPERA I) (n=821) | WA21093 (OPERA II) (n=835) | ||
| Study design | ||||
| Study population | Patients with relapsing forms of MS | |||
| Disease history at screening | At least two relapses within the prior two years or one relapse within the prior year; EDSS* between 0 and 5.5, inclusive | |||
| Study duration | 2 years | |||
| Treatment groups | Group A: Ocrevus 600 mg | |||
| Group B: interferon beta-1a 44 mcg S.C. (IFN) | ||||
| Baseline characteristics | Ocrevus 600 mg (n=410) | IFN 44 mcg (n=411) | Ocrevus 600 mg (n=417) | IFN 44 mcg (n=418) |
| Mean age (years) | 37.1 | 36.9 | 37.2 | 37.4 |
| Age range (years) at inclusion | 18–56 | 18-55 | 18-55 | 18-55 |
| Gender distribution (% male/% female) | 34.1/65.9 | 33.8/66.2 | 35.0/65.0 | 33.0/67.0 |
| Mean/Median disease duration since diagnosis (years) | 3.82/1.53 | 3.71/1.57 | 4.15/2.10 | 4.13/1.84 |
| Patients naive to previous DMT (%)** | 73.4 | 71.0 | 72.7 | 74.9 |
| Mean number of relapses in the last year | 1.31 | 1.33 | 1.32 | 1.34 |
| Proportion of patients with Gd enhancing T1 lesions | 42.5 | 38.1 | 39.0 | 41.4 |
| Mean EDSS* | 2.82 | 2.71 | 2.73 | 2.79 |
* Expanded Disability Status Scale
** Patients who had not been treated with any MS medication in the 2 years prior to randomization.
Key clinical and MRI efficacy results are presented in Table 4 and Figure 1.
The results of these studies show that Ocrevus significantly suppressed relapses, sub-clinical disease activity measured by MRI, and disease progression compared with interferon beta-1a 44mcg subcutaneous.
Table 4. Key Clinical and MRI Endpoints from Studies WA21092 and WA21093 (RMS):
| Endpoints | Study 1: WA21092 (OPERA I) | Study 2: WA21093 (OPERA II) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Ocrevus 600 mg (n=410) | IFN 44 mcg (n=411) | Ocrevus 600 mg (n=417) | IFN 44 mcg (n=418) | ||||||
| Clinical Endpoints | |||||||||
| Annualised Relapse Rate (ARR) (primary endpoint) | 0.156 | 0.292 | 0.155 | 0.290 | |||||
| Relative Reduction | 46% (p<0.0001) | 47% (p<0.0001) | |||||||
| Proportion of patients with 12 week Confirmed Disability Progression3 | 9.8% Ocrevus έναντι 15.2% IFN | ||||||||
| Risk Reduction (Pooled Analysis1) | 40% (p=0,0006)7 | ||||||||
| Risk Reduction (Individual Studies2) | 43% (p=0.0139)7 | 37% (p=0.0169)7 | |||||||
| Proportion of patients with 24 week Confirmed Disability Progression3 | 7,6% Ocrevus vs 12.0% IFN | ||||||||
| Risk Reduction (Pooled Analysis1) | 40% (p=0.0025)7 | ||||||||
| Risk Reduction (Individual Studies2) | 43% (p=0.0278)7 | 37% (p=0.0370)7 | |||||||
| Proportion of patients with at least 12 weeks Confirmed Disability Improvement4 | 20.7% Ocrevus έναντι 15.6% IFN | ||||||||
| Relative Increase (Pooled Analysis1) | 33% (p=0.0194) | ||||||||
| Relative Increase (Individual Studies2) | 61% (p=0,0106) | 14% (p=0,4019) | |||||||
| Proportion of patients Relapse free at 96 weeks2 | 80.4% | 66.7% | 78.9% | 64.3% | |||||
| (p<0.0001) | (p<0.0001) | ||||||||
| Proportion of patients with No Evidence of Disease Activity (NEDA)5 | 48% | 29% | 48% | 25% | |||||
| Relative Increase2 | 64% (p<0.0001) | 89% (p<0.0001) | |||||||
| MRI Endpoints | |||||||||
| Mean number of T1 Gd-enhancing lesions per MRI scan | 0.016 | 0.286 | 0.021 | 0.416 | |||||
| Relative reduction | 94% (p<0,0001) | 95% (p<0,0001) | |||||||
| Mean number of new and/or enlarging T2 hyperintense lesions per MRI scan | 0.323 | 1.413 | 0.325 | 1.904 | |||||
| Relative reduction | 77% (p<0.0001) | 83% (p<0.0001) | Percentage change in brain volume from Week 24 to week 96 | -0.572 | -0.741 | -0.638 | -0.750 | ||
| Relative reduction in brain volume loss | 22.8% (p=0.0042)6 | 14.9% (p=0.0900) | |||||||
1 Data prospectively pooled from Study 1 and 2
2 Non-confirmatory p-value analysis; not part of the pre-specified testing hierarchy
3 Defined as an increase of ≥1.0 point from the baseline Expanded Disability Status Scale (EDSS) score for patients with baseline score of 5.5 or less, or ≥0.5 when the baseline score is >5.5, Kaplan-Meier estimates at Week 96
4 Defined as decrease of ≥1.0 point from the baseline EDSS score for patients with baseline EDSS score ≥2 and ≤5.5, or ≥0.5 when the baseline score is >5.5. Patients with baseline score <2 were not included in analysis.
5 NEDA defined as absence of protocol defined relapses, 12-week Confirmed Disability Progression (CDP), and any MRI activity (either Gd-enhancing T1 lesions, or new or enlarging T2 lesions) during the whole 96-week treatment. Exploratory result based on complete ITT population.
6 Non-confirmatory p-value; hierarchical testing procedure terminated before reaching endpoint.
7 Log-rank test
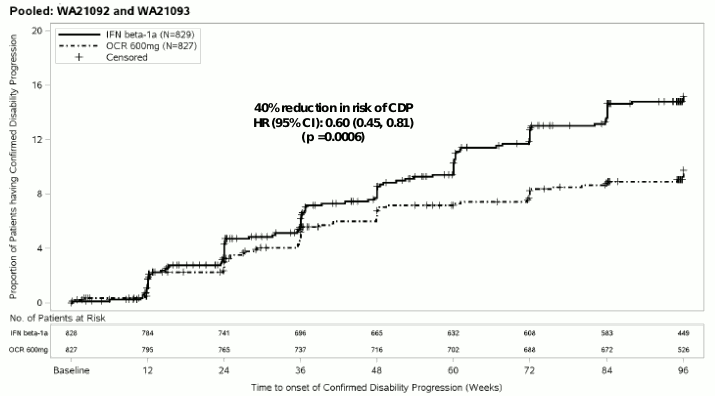
Figure 1. Kaplan-Meier Plot of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks with the Initial Event of Neurological Worsening Occurring during the Double-blind Treatment Period (Pooled WA21092 and WA21093 ITT Population)*:
* Pre-specified pooled analysis of WA21092 and WA21093
Results of the pre-specified pooled analyses of time to CDP sustained for at least 12 weeks (40% risk reduction for Ocrevus compared to interferon beta-1a (p=0.0006) were highly consistent with the results sustained for at least 24 weeks (40% risk reduction for Ocrevus compared to interferon beta-1a, p=0.0025).
The studies enrolled patients with active disease. These included both active treatment naive and previously treated inadequate responders, as defined by clinical or imaging features. Analysis of patient populations with differing baseline levels of disease activity, including active and highly active disease, showed that the efficacy of Ocrevus on ARR and 12 week CDP was consistent with the overall population.
Primary Progressive MS
Efficacy and safety of Ocrevus were also evaluated in a randomised, double-blind, placebo-controlled clinical trial in patients with primary progressive MS (Study WA25046) who were early in their disease course according to the main inclusion criteria, i.e. ages 18-55 years, inclusive; EDSS at screening from 3.0 to 6.5 points; disease duration from the onset of MS symptoms less than 10 years in patients with an EDSS at screening ≤5.0 or less than 15 years in patients with an EDSS at screening >5.0. With regard to disease activity, features characteristic of inflammatory activity, even in progressive MS, can be imaging-related, (i.e. T1 Gd-enhancing lesions and/or active [new or enlarging] T2 lesions). MRI evidence should be used to confirm inflammatory activity in all patients. Patients over 55 years of age were not studied. Study design and baseline characteristics of the study population are presented in Table 5.
Demographic and baseline characteristics were well balanced across the two treatment groups. Cranial MRI showed imaging features characteristic of inflammatory activity either by T1 Gd enhancing lesions or T2 lesions.
During the Phase 3 PPMS study, patients received 600 mg Ocrevus every 6 months as two 300 mg infusions, given two weeks apart, throughout the treatment period. The 600 mg infusions in RMS and the 2 x 300 mg infusions in PPMS demonstrated consistent PK/PD profiles. IRR profiles per infusion were also similar, independent of whether the 600 mg dose was administered as a single 600 mg infusion or as two 300 mg infusions separated by two weeks (see sections 4.8 and 5.2), but due to overall more infusions with the 2 x 300 mg regimen, the total number of IRRs were higher. Therefore, after Dose 1 it is recommended to administer Ocrevus in a 600 mg single infusion (see section 4.2) to reduce the total number of infusions (with concurrent exposure to prophylactic methylprednisolone and an antihistamine) and the related infusion reactions).
Table 5. Study design, demograhics and baseline characteristics for Study WA25046:
| Study name | Study WA25046 ORATORIO (n=732) | |
|---|---|---|
| Study design | ||
| Study population | Patients with primary progressive form of MS | |
| Study duration | Event-driven (Minimum 120 weeks and 253 confirmed disability progression events) (Median follow-up time: Ocrevus 3.0 years, Placebo 2.8 years | |
| Disease history at screening | Age 18-55 years, EDSS of 3.0 to 6.5 | |
| Treatment groups | Group A: Ocrevus 600 mg | |
| Group B: Placebo, in 2:1 randomisation | ||
| Baseline characteristics | Ocrevus 600 mg (n=488) | Placebo (n=244) |
| Mean age (years) | 44.7 | 44.4 |
| Age range (years) at inclusion | 20-56 | 18-56 |
| Gender distribution (% male/% female) | 51.4/48.6 | 49.2/50.8 |
| Mean/Median disease duration since PPMS diagnosis (years) | 2.9/1.6 | 2.8/1.3 |
| Mean EDSS | 4.7 | 4.7 |
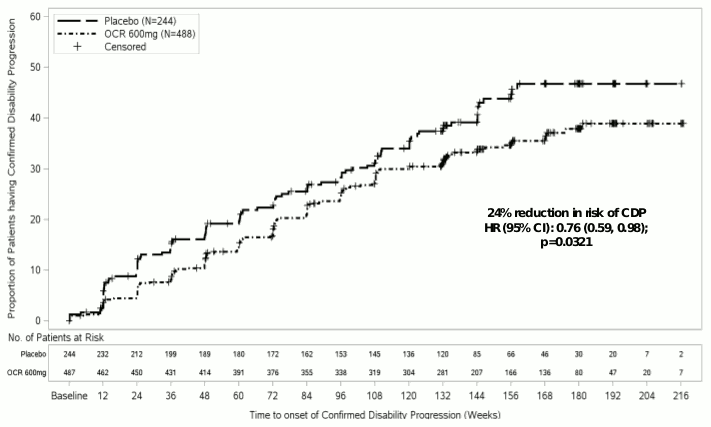
Key clinical and MRI efficacy results are presented in Table 6 and Figure 2.
The results of this study show that Ocrevus significantly delays disease progression and reduces deterioration in walking speed compared with placebo.
Table 6. Key Clinical and MRI Endpoints from Study WA25046 (PPMS):
| Study 3 | ||
|---|---|---|
| WA25046 (Oratorio) | ||
| Endpoints | Ocrevus 600 mg (n=488) | Placebo (n=244) |
| Clinical Endpoints | ||
| Primary efficacy endpoint | ||
| Proportion of patients with 12 weeks - Confirmed Disability Progression1 (primary endpoint) | 30.2% | 34.0% |
| Risk reduction | 24% (p=0.0321) | |
| Proportion of patients with 24 weeks - Confirmed Disability Progression1 | 28.3% | 32.7% |
| Risk reduction | 25% (p=0.0365) | |
| Percentage change in Timed 25-Foot Walk from baseline to Week 120 | 38.9 | 55.1 |
| Relative reduction in progression rate of walking time | 29.4% (p=0.0404) | |
| MRI Endpoints | ||
| Percentage change in T2 hyperintense lesion volume, from baseline to Week 120 | -3.4 | 7.4 |
| (p<0.0001) | ||
| Percentage change in brain volume from Week 24 to Week 120 | -0,902 | -1.093 |
| Relative reduction in rate of brain volume loss | 17.5% (p=0.0206) | |
1 Defined as an increase of ≥1.0 point from the baseline EDSS score for patients with baseline score of 5.5 or less, or ≥0.5 when the baseline score is >5.5, Kaplan-Meier estimates at Week 120.
Figure 2: Kaplan-Meier Plot of Time to Onset of Confirmed Disability Progression Sustained for at Least 12 Weeks with the Initial Event of Neurological Worsening Occurring during the Double-blind Treatment Period (WA25046 ITT Population)*:
* All patients in this analysis had a minimum of 120 weeks of follow-up. The primary analysis is based on all events accrued.
Pre-specified non-powered subgroup analysis of the the primary endpoint suggests that patients who are younger or those with T1 Gd-enhancing lesions at baseline receive a greater treatment benefit than patients who are older or without T1 Gd-enhancing lesions (≤45 years: HR 0.64 [0.45, 0.92], >45 years: HR 0.88 [0.62, 1.26]; with T1 Gd-enhancing lesions at baseline: HR 0.65 [0.40-1.06], without T1 Gd-enhancing lesions at baseline: HR 0.84 [0.62-1.13]). Moreover, post-hoc analyses suggested that younger patients with T1 Gd-enhancing lesions at baseline have the better treatment effect (≤45 years: HR 0.52 [0.27-1.00]; ≤46 years [median age of the WA25046 study]; HR 0.48 [0.25-0.92]; <51 years: HR 0.53 [0.31-0.89]).
Immunogenicity
Patients in MS trials (WA21092, WA21093 and WA25046) were tested at multiple time points (baseline and every 6 months post treatment for the duration of the trial) for anti-drug antibodies (ADAs). Out of 1311 patients treated with Ocrevus, 12 (~1%) tested positive for treatment-emergent ADAs, of which 2 patients tested positive for neutralising antibodies. The impact of treatment-emergent ADAs on safety and efficacy cannot be assessed given the low incidence of ADA associated with Ocrevus.
Immunisations
In a randomized open-label study in RMS patients (N=102), the percentage of patients with a positive response to tetanus vaccine at 8 weeks after vaccination was 23.9% in the ocrelizumab group compared to 54.5% in the control group (no disease-modifying therapy except interferon-beta). Geometric mean anti-tetanus toxoid specific antibody titers at 8 weeks were 3.74 and 9.81 IU/ml, respectively. Positive response to 35 serotypes in 23-PPV at 4 weeks after vaccination was 71.6% in the ocrelizumab group and 100% in the control group. In patients treated with ocrelizumab a booster vaccine (13-PCV) given 4 weeks after 23-PPV did not markedly enhance the response to 12 serotypes in common with 23-PPV. The percentage of patients with seroprotective titers against five influenza strains ranged from 20.0-60.0% and 16.7-43.8% pre-vaccination and at 4 weeks post vaccination from 55.6-80.0% in patients treated with ocrelizumab and 75.0-97.0% in the control group, respectively. See sections 4.4 and 4.5.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with Ocrevus in one or more subsets of the paediatric population in the treatment of multiple sclerosis. See section 4.2 for information on paediatric use.
Pharmacokinetic properties
The pharmacokinetics of ocrelizumab in the MS studies were described by a two compartment model with time-dependent clearance, and with PK parameters typical for an IgG1 monoclonal antibody. The overall exposure (AUC over the 24 weeks dosing interval) was identical in the 2 x 300 mg in PPMS and 1 x 600 mg in RMS studies, as expected given an identical dose was administered. Area under the curve (AUCτ) after the 4th dose of 600 mg ocrelizumab was 3510 μg/mL•day, and mean maximum concentration (Cmax) was 212 μg/mL in RMS (600 mg infusion) and 141 μg/mL in PPMS (300 mg infusions).
Absorption
Ocrevus is administered as an intravenous infusion. There have been no studies performed with other routes of administration.
Distribution
The population pharmacokinetics estimate of the central volume of distribution was 2.78 L. Peripheral volume and inter-compartment clearance were estimated at 2.68 L and 0.294 L/day.
Biotransformation
The metabolism of Ocrevus has not been directly studied, as antibodies are cleared principally by catabolism (i.e. breakdown into peptides and amino acids).
Elimination
Constant clearance was estimated at 0.17 L/day, and initial time-dependent clearance at 0.0489 L/day which declined with a half-life of 33 weeks. The terminal elimination half-life of ocrelizumab was 26 days.
Pharmacokinetics in Special Populations
Paediatrics
No studies have been conducted to investigate the pharmacokinetics of ocrelizumab in children and adolescents <18 years of age.
Elderly
There are no dedicated PK studies of ocrelizumab in patients ≥55 years due to limited clinical experience (see section 4.2).
Renal impairment
No formal pharmacokinetic study has been conducted. Patients with mild renal impairment were included in clinical trials and no change in the pharmacokinetics of Ocrevus was observed in those patients. There is no PK information available in patients with moderate or severe renal impairment.
Hepatic impairment
No formal pharmacokinetic study has been conducted. Patients with mild hepatic impairment were included in clinical trials, and no change in the pharmacokinetics was observed in those patients. There is no PK information available in patients with moderate or severe hepatic impairment.
Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, repeated dose toxicity, and embryo-foetal development. Neither carcinogenicity nor mutagenicity studies have been conducted with ocrelizumab.
In a pre- and post-natal development study in cynomolgus monkeys, administration of ocrelizumab from gestation day 20 to approximately 5 weeks postpartum was associated with glomerulopathy, lymphoid follicle formation in bone marrow, lymphoplasmacytic renal inflammation, and decreased testicular weight in offspring. The maternal doses administered in this study resulted in maximum mean serum concentrations (Cmax) that were 4.5- and 21-fold above those anticipated in the clinical setting.
There were two cases of moribundityone attributed to weakness due to premature birth accompanied by opportunistic infection and the other to an infective meningoencephalitis involving the cerebellum of the neonate from a maternal dam with an active infection (mastitis). The course of both neonatal infections could have potentially been impacted by B-cell depletion. Newborn offspring of maternal animals exposed to ocrelizumab were noted to have depleted B cell populations during the post natal phase. Measurable levels of ocrelizumab were detected in milk (approximated 0.2% of steady state trough serum levels) during the lactation period.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.