ONTOZRY Tablet / Film-coated tablet Ref.[49751] Active ingredients: Cenobamate
Source: European Medicines Agency (EU) Revision Year: 2022 Publisher: Angelini Pharma S.p.A, Viale Amelia 70, 00181, Rome – Italy
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: antiepileptics, other antiepileptics
ATC code: N03AX25
Mechanism of action
Cenobamate is a small molecule with a dual mechanism of action. It is a positive allosteric modulator of subtypes of the γ-aminobutyric acid (GABAA) ion channel, that does not bind to the benzodiazepine binding site. Cenobamate has also been shown to reduce repetitive neuronal firing by enhancing the inactivation of sodium channels and by inhibiting the persistent component of the sodium current. The precise mechanism of action by which cenobamate exercises its therapeutic effectsin patients with focalonset seizures is unknown.
Pharmacodynamic effects
Cardiac electrophysiology
In a placebo-controlled QT study in healthy volunteers, dose-dependent shortening of the QTcF interval has been observed with cenobamate. The mean ΔΔQTcF is -10.8 [CI: -13.4, -8.2] msec for 200 mg once daily and -18.4 [CI: -21.5, -15.2] msec for 500 mg once daily (1.25 times the maximum recommended dosage). Reductions of the QTc interval below 340 msec were not observed (see section 4.4).
Clinical efficacy and safety
The efficacy of cenobamate as adjunctive therapy in focal-onset seizures was studied in a multi-centre, randomised, double-blind, placebo-controlled study in adult patients with focal-onset epilepsy who have not been adequately controlled despite a history of treatment with anti-epileptic products. Patients were treated with one to three concomitant antiepileptic medicinal products that remained stable over the course of double-blind study treatment. The daily dose of cenobamate ranged from 100 to 400 mg/day.
The study had an 8-week prospective baseline period, during which patients were required to have at least 3 or 4 partial-onset seizures per 28 days with no seizure-free period exceeding 3 to 4 weeks, followed by an 18-week treatment period including 12 weeks at fixed. The most commonly taken antiepileptic medicinal products at the time of study entry were levetiracetam, lamotrigine, carbamazepine and lacosamide. All subjects who entered the study continued to have seizures, despite a majority having had a history of treatment with 2 or more antiepileptic medicinal products. More than 80% of patients were taking two or more concomitant antiepileptic medicinal products at the time of study enrolment. The efficacy outcomes are summarised in table 3.
The study compared doses of cenobamate 100 mg/day, 200 mg/day and 400 mg/day with placebo, on top of standard of care. Subjects continued stable treatment on one to three background antiepileptic medicinal products. Patients were started on a daily dose of 50 mg and subsequently increased by 50 mg/day every week until 200 mg/day was reached and then increased by 100 mg/day every week in subjects randomised to 400 mg/day.
Table 3 shows the proportion of patients who exhibited a 50% or greater reduction in seizure frequency from baseline.
Table 3. Proportion of patients exhibiting 50% or greater response in Study C017:
| Study | Standard of care and placebo | Standard of care and cenobamate | ||
|---|---|---|---|---|
| 100 mg/day | 200 mg/day | 400 mg/day | ||
| Study C017 | ||||
| n=102 | n=102 | n=98 | n=95 | |
| 50% Responder rate1 | 26 (25.5%) | 41 (40.2%) | 55 (56.1%) | 61 (64.2%) |
| Cenobamate placebo difference | 14.7% (p=0.036) | 30.6% (p<0.001) | 38.7% (p<0.001) | |
1 Over 12 weeks of fixed-dose double-blind treatment.
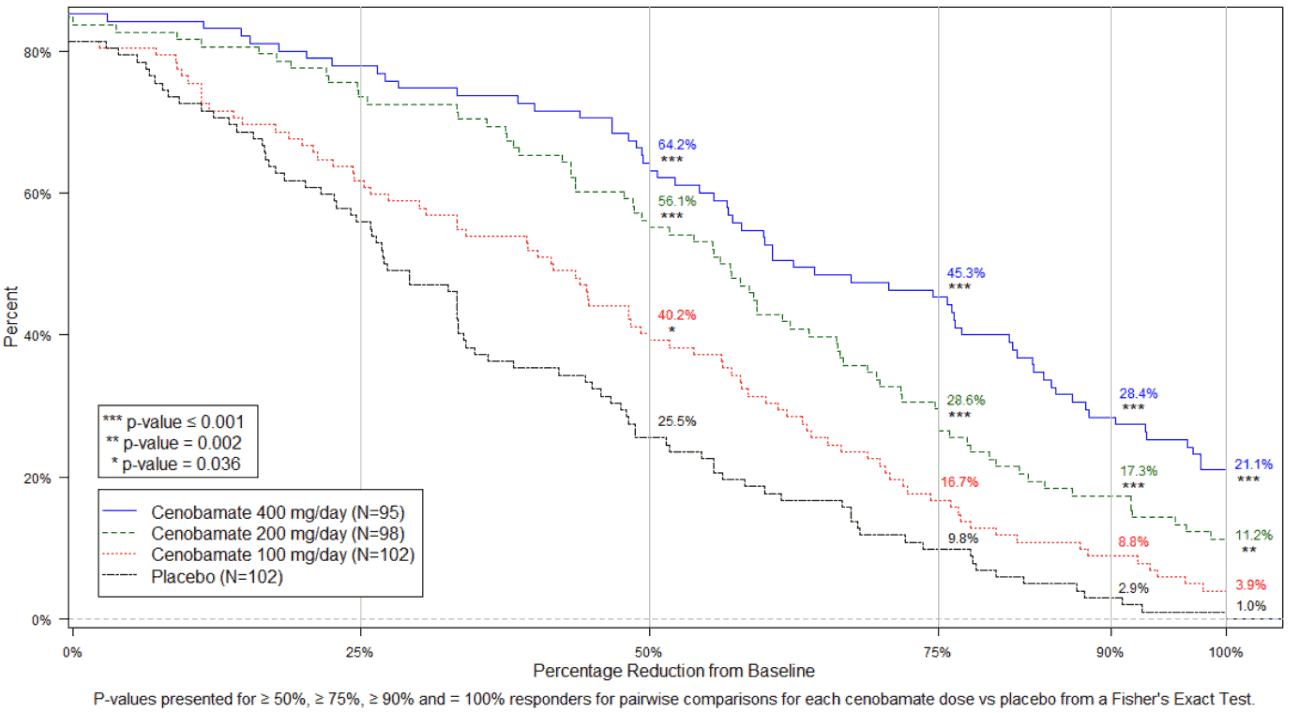
Figure 1 shows the percentage of patients by category of seizure response during the maintenance phase with increasingly stringent criteria for response.
Figure 1. Cumulative distribution of percent reduction in seizures from baseline by treatment group in the 12-week fixed-dose period in the Study:
In the study, 4 of 102 (3.9%) patients in the cenobamate 100 mg/day group, 11 of 98 (11.2%) patients in the cenobamate 200 mg/day group, 20 of 95 (21.1%) patients in the cenobamate 400 mg/day group and 1 of 102 (1%) of patients in the placebo group obtained seizure freedom (100% reduction in seizures) during the 12-week fixed-dose phase. Similar responses were seen across subpopulations greater than or less than median seizure frequency, and greater than or less than median disease duration.
Long term open label study
The majority of subjects chose to enter the open-label extension from Study 1 (98.9%). 80% of subjects remained in the study for at least 12 months, and 58% for at least 60 months. Additional seizure frequency data were collected and were consistent with the results from the double-blind portion of the study.
Paediatric population
The European Medicines Agency has deferred the obligation to submit the results of studies with Ontozry in one or more subsets of the paediatric population in epilepsy (see section 4.2 for information on paediatric use).
5.2. Pharmacokinetic properties
Absorption
Cenobamate is well absorbed (at least 88% based on urine recovery) after oral administration, with median Tmax ranging from 1 to 4 hours after single- or multiple-dose administration under fasted condition over the range of 10 to 400 mg. Co-administration with a high-fat meal (800-1,000 kcal with 50% fat) showed no significant effect on the rate and the extent of absorption of cenobamate.
Distribution
The apparent volume of distribution (Vd/F) of cenobamate after oral administration is approximately 40-50 L. Plasma protein binding of cenobamate is 60% and independent of concentration in vitro. Cenobamate primarily binds with human albumin protein.
Biotransformation
Cenobamate is extensively metabolised. The primary metabolic pathway is glucuronidation via UGT2B7 and to a lesser extent by UGT2B4. Minor pathways for metabolism of cenobamate include oxidation via CYP2E1, CYP2A6, CYP2B6, and to a lesser extent by CYP2C19 and CYP3A4/5.
Elimination
Cenobamate and its metabolites are eliminated primarily via urine. Excretion via faeces accounted for only 5.2% of the dose. More than 50% of the dose was excreted within 72 hours. The apparent terminal half-life of cenobamate in plasma was 50-60 hours within the therapeutic range of 100 mg/day to 400 mg/day. Steady state is reached by 14 days.
Linearity/non-linearity
The Cmax of cenobamate increased proportionally with increasing doses following single oral doses from 5 to 750 mg and multiple oral doses from 50 to 500 mg/day. Steady-state exposures (Cmax and AUC) increased proportionally with increasing doses in the therapeutic range (100 to 400 mg), but doses less than 100 mg/day may be cleared faster.
Special populations
Renal impairment
Cenobamate plasma AUC was 1.4-fold to 1.5-fold higher in subjects with mild (CLcr 60 to <90 mL/min) and moderate (CLcr 30 to <60 mL/min) renal impairment following a single oral 200 mg dose of cenobamate compared to healthy controls. In subjects with severe (CLcr <30 mL/min) renal impairment, cenobamate plasma AUC did not change significantly compared to healthy controls following single oral 100 mg dose of cenobamate (see section 4.2), The effect of haemodialysis on cenobamate pharmacokinetics has not been studied.
Hepatic impairment
Cenobamate plasma AUC was 1.9-fold and 2.3-fold higher in subjects with mild and moderate hepatic impairment, respectively, following a single oral 200 mg dose of cenobamate compared to matched healthy controls (see section 4.2). The effect of severe hepatic impairment on cenobamate pharmacokinetics has not been studied.
Gender
There was no difference observed in the pharmacokinetics of cenobamate between male and female patients.
Ethnicity
No clinically significant effect of ethnicity on the pharmacokinetics of cenobamate was noted in a population PK analysis of pooled data from clinical studies from subjects categorised as Asian, Black, Caucasian, Hispanic or other.
Body weight
A 45% decrease in exposure has been estimated across a body weight range from 54 kg to 112 kg. This variability is not considered to be clinically relevant when establishing a dose of cenobamate. However, cenobamate dose adjustments may need to be considered in patients who experience weight changes of ≥30% of their initial body weight, or more.
Elderly (65 years and above)
No clinically significant differences in the pharmacokinetics of cenobamate were observed based on age based on data from subjects aged 18 years to 77 years.
Paediatric population
Safety and effectiveness of Ontozry in patients less than 18 years of age has not been established.
5.3. Preclinical safety data
Non-clinical data reveal no special hazard for humans based on conventional studies of safety pharmacology, genotoxicity, and carcinogenic potential. However, maximum systemic exposure achieved in the carcinogenicity study in rats was less than that in humans at the maximum recommended human dose (MRHD) of 400 mg/day.
Repeated dose toxicity
Maximum doses in repeat dose toxicity studies were limited by the exaggerated CNS effects of cenobamate (including hypoactivity, uncoordinated gait, hypothermia, and tremor). Systemic exposures at NOAEL (no observed adverse effect levels) were identified or below exposures reached in humans at the MRHD.
Toxicity to reproduction and development
Reproductive toxicity studies showed adverse effects on embryo-foetal and postnatal development. No adverse effects were observed on fertility. However, systemic exposures at the respective NOAELs for the fertility, embryo-foetal development and pre-postnatal development were bellow human exposure at the MRHD.
Administration of cenobamate to pregnant rats and rabbits during the period of organogenesis resulted in increased embryo-foetal mortality, at dose levels associated with maternal toxicity. In rats, there was a small increase in visceral malformations at the high dose; however full interpretation of the teratogenic potential at the high dose was not possible due to the high maternal toxicity.
When cenobamate was administered to female rats throughout pregnancy and lactation, neurobehavioural impairment (increased auditory startle response) was observed in the offspring at all doses and decreased preweaning body weight gain and adverse reactions on female reproductive function (decreased numbers of corpora lutea, implantations and live foetuses) were seen in the offspring
Placental and lacteal transfer of cenobamate was confirmed by the presence of cenobamate in both amniotic fluid and foetal blood from pregnant rats and in the milk of lactating rats.
The environmental risk assessment demonstrated that cenobamate is very persistent (vP) in aquatic systems (see section 6.6).
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.