PARMODIA Film-coated tablet Ref.[51004] Active ingredients: Pemafibrate
Source: Health Sciences Authority (SG) Revision Year: 2022 Publisher: Kowa Company, Ltd., 6-29, Nishiki 3-chome, Naka-ku, Nagoya, Aichi, JAPAN
5.1. Pharmacodynamic properties
Pharmacotherapeutic group: Not yet assigned
ATC Code: Not yet assigned
Mechanism of action
Pemafibrate activates PPARα by binding to this receptor and regulates the target gene expression, leading to decreased plasma triglyceride (TG) concentration, decreased triglyceride-rich lipoprotein, decreased apolipoprotein (Apo) C-3, and increased HDL-cholesterol.
(1) The activation of PPARα by pemafibrate was more potent than the activation of PPARγ or PPARδ, indicating the selectivity of pemafibrate to PPARα (in vitro).
(2) Pemafibrate inhibited TG synthesis in the liver (rats).
(3) Pemafibrate significantly reduced TG secretory rate (rats).
(4) Pemafibrate increased LPL activity (rats).
(5) Pemafibrate significantly reduced plasma concentrations of ApoC-3 and Angiopoietin-like Protein 3, which negatively regulate LPL activity; moreover, pemafibrate inhibited the gene expression (Apoc3, Angptl3) in the liver. In addition, pemafibrate upregulated the expression of genes (Aco, Cpt1a) involved in β-oxidation of free fatty acids that inhibits LPL activity (rats).
(6) Pemafibrate facilitated plasma TG clearance (rats).
(7) Pemafibrate increased plasma concentration of fibroblast growth factor 21 (FGF21), a protein that reduces TG concentration and increases HDLcholesterol concentration (rats).
Pharmacodynamic effects
Pharmacological action
(1) Effect of reducing plasma lipid
When pemafibrate was orally administered to rats with high fructose-induced hypertriglyceridemia, plasma TG concentration was decreased in a dosedependent manner.
(2) Effect of increasing HDL-cholesterol
When pemafibrate was orally administered to human ApoA-1 transgenic mice, plasma concentration of HDL-cholesterol and concentration of human ApoA-1 were increased.
(3) Anti-arteriosclerotic effect
When pemafibrate was orally administered to LDL-receptor deficient mice under high fat/high cholesterol diet, the area of lipid deposition area in the aortic sinus was decreased.
Clinical efficacy
Phase ⅔ Comparative Confirmatory Study with Fenofibrate
In patients with dyslipidemia who had high TG and low HDL-cholesterol levels, placebo, 0.2 mg/day or 0.4 mg/day of PARMODIA (twice daily after breakfast and dinner), or micronized fenofibrate capsules of 100 mg/day or 200 mg/day (once daily after breakfast) was administered for 12 weeks. The percent change in fasting serum TG was as presented in the following table, which shows the superiority of PARMODIA groups over the placebo group, and non-inferiority of PARMODIA 0.2 mg/day and 0.4 mg/day groups over the micronized fenofibrate capsule 200 mg/day group.
Table 1. Percent change in fasting serum TG in placebo group and PARMODIA groups:
| Treatment group and Baseline fasting serum TGa (mg/dL) | Percent change in fasting serum TGb | |
|---|---|---|
| Percent change from baselinec (%) | Difference from placebo in percent changed) (%) | |
| Placebo 346.1±130.9, n=43 | -2.775 [-11.783, 6.233] | - |
| PARMODIA 0.2 mg/day 367.2±153.6, n=128 | -46.766 [-49.985, -43.547] | -43.991** [-55.455, -32.528] |
| PARMODIA 0.4 mg/day 362.6±158.5, n=84 | -51.902 [-55.841, -47.963] | -49.127** [-60.922, -37.333] |
a Mean ± SD To convert TG from mg/dL to mmol/L, multiply by 0.0113
b Repeated measures analysis of covariance for all treatment groups, with Weeks 8, 10, and 12 as repeated time points and baseline value as a covariate (The results of the PARMODIA 0.1 mg/day group are omitted.)
c Least square mean [95% CI]
d Least square mean [Adjusted 95% CI]
**: p ≤0.01 (Dunnett’s test)
Table 2. Percent change in fasting serum TG in PARMODIA groups and micronized fenofibrate capsule groups:
| Treatment group and Baseline fasting serum TGa (mg/dL) | Percent change in fasting serum TGb | |
|---|---|---|
| Percent change from baseline (%) | Difference from micronized fenofibrate capsule 200 mg/day group in percent change (%) | |
| PARMODIA 0.2 mg/day 367.2±153.6, n=128 | -46.690 [-49.904, -43.477] | 4.844 [0.388, 9.299] |
| PARMODIA 0.4 mg/day 362.6±158.5, n=84 | -51.836 [-55.768, -47.903] | -0.302 [-5.300, 4.696] |
| Micronized fenofibrate capsule 100 mg/dayc 362.0±135.1, n=85 | -38.261 [-42.230, -34.291] | - |
| Micronized fenofibrate capsule 200 mg/dayc 347.3±123.8, n=140 | -51.534 [-54.616, -48.452] | - |
a Mean ± SD To convert TG from mg/dL to mmol/L, multiply by 0.0113
b Repeated measures analysis of covariance for all treatment groups, with Weeks
8, 10, and 12 as repeated time points and baseline value as a covariate (The results of the PARMODIA 0.1 mg/day group are omitted.)
Least square mean [95% CI] Non-inferiority margin: 10%
The change over time in LDL-cholesterol was as presented in the following table.
Table 3. Change over time in LDL-cholesterol by group:
| Placebo group | PARMODIA group | Micronized fenofibrate capsule group | |||
|---|---|---|---|---|---|
| 0.2 mg/day | 0.4 mg/day | 100 mg/day | 200 mg/day | ||
| Baseline | 133.8±33.9 (43) | 131.4±35.5 (128) | 125.9±33.5 (84) | 133.8±35.9 (85) | 133.8±36.1 (140) |
| Week 4 | 130.2±32.0 (43) | 143.2±33.0 (127) | 139.5±29.6 (83) | 142.2±34.1 (83) | 136.5±30.5 (139) |
| Week 8 | 137.8±32.3 (43) | 147.8±35.7 (124) | 141.7±30.6 (83) | 148.2±32.6 (81) | 135.8±30.9 (136) |
| Week 12 | 131.8±33.3 (43) | 149.1±33.3 (122) | 144.8±32.2 (80) | 148.8±32.5 (79) | 137.0±32.3 (128) |
Mean ± SD (mg/dL)
To convert LDL-C from mg/dL to mmol/L, multiply by 0.0259
(number of subjects)
Phase 3 Comparative Confirmatory Study with Fenofibrate
In patients with dyslipidemia who had high TG and low HDL-cholesterol levels, placebo, 0.2 mg/day or 0.4 mg/day of PARMODIA (twice daily after breakfast and dinner), or fenofibrate tablets of 106.6 mg/day (once daily after breakfast) was administered for 24 weeks. The fenofibrate tablets (solid dispersion) of 106.6 mg are equivalent to micronized fenofibrate capsules of 134 mg. The percent change in fasting serum TG was as presented in the following table, which shows the noninferiority of all PARMODIA groups over the fenofibrate tablet 106.6 mg/day group.
Table 4. Percent change in fasting serum TG in PARMODIA groups and fenofibrate tablet group:
| Treatment group and Baseline fasting serum TGa) (mg/dL) | Percent change in fasting serum TGb | |
|---|---|---|
| Percent change from baseline (%) | Difference from fenofibrate tablet 106.6 mg/day groupc in percent change | |
| PARMODIA 0.2 mg/day 242.4±53.3, n=73 | -46.226 [-50.122, -42.329] | -6.541 [-12.004, -1.078] |
| PARMODIA 0.4 mg/day 233.3±60.8, n=74 | -45.850 [-49.678, -42.023] | -6.166 [-11.576, -0.755] |
| Fenofibrate tablet 106.6mg/day 235.6±71.7, n=76 | -39.685 [-43.511, -35.858] | - |
a Mean ± SD To convert TG from mg/dL to mmol/L, multiply by 0.0113
b Repeated measures analysis of covariance with Weeks 8, 12, 16, 20, and 24 as repeated time points and baseline value as a covariate Least square mean [95% CI] Non-inferiority margin: 10%
c Fenofibrate tablets (solid dispersion) of 106.6 mg are equivalent to micronized fenofibrate capsules of 134 mg.
The change over time in the LDL-cholesterol was as presented in the following table.
Table 5. Change over time in LDL-cholesterol by group:
| PARMODIA group | Fenofibrate tablet 106.6 mg/day group | ||
|---|---|---|---|
| 0.2 mg/day | 0.4 mg/day | ||
| Baseline | 157.8±29.2 (73) | 154.0±27.4 (74) | 152.6±26.1 (76) |
| Week 4 | 145.4±23.0 (73) | 144.2±30.6 (74) | 142.8±27.2 (76) |
| Week 8 | 145.4±24.6 (72) | 145.7±32.3 (74) | 139.7±28.8 (76) |
| Week 12 | 146.3±23.9 (71) | 144.0±33.4 (74) | 143.6±27.9 (72) |
| Week 16 | 144.4±25.0 (71) | 142.0±33.0 (74) | 138.8±30.0 (71) |
| Week 20 | 145.1±21.5 (70) | 143.1±31.5 (74) | 139.0±29.4 (70) |
| Week 24 | 144.6±26.5 (69) | 147.0±32.2 (73) | 141.4±31.7 (68) |
| Week 24 (LOCF) | 144.7±25.8 (73) | 146.7±32.0 (74) | 142.2±31.5 (76) |
Mean ± SD (mg/dL)
To convert LDL-C from mg/dL to mmol/L, multiply by 0.0259
(number of subjects)
LOCF: Last observation carried forward
Phase 3 Long-term Administration Study in Dyslipidemia Patients with High TG Levels
In patients with dyslipidemia who had high TG levels, PARMODIA 0.2 mg/day (a dose increase to PARMODIA 0.4 mg/day was allowed as necessary in subjects with inadequate response to PARMODIA 0.2 mg/day at Week 12 and after) was administered twice daily before or after breakfast and dinner for 52 weeks. The percent change from the baseline fasting serum TG of 249.7±77.5 mg/dL (2.82±0.88 mmol/L) (Mean ± SD [the same applies hereinafter], n=189) at Week 24 and Week 52 were -48.77±20.47% and -45.93±21.84%, respectively (Last observation carried forward [LOCF] method was used). LDL-cholesterol value was 119.3±31.7 mg/dL (3.09±0.82 mmol/L) at baseline, and 116.6±29.1 mg/dL (3.02±0.75 mmol/L) at Week 52 (n=189).
Phase 3 Long-term Administration Study in Patients with Dyslipidemia and Type 2 Diabetes Mellitus
In patients with dyslipidemia and type 2 diabetes mellitus, placebo/PARMODIA 0.2 mg/day (starting from Week 24, the treatment was switched from placebo to PARMODIA 0.2 mg/day), PARMODIA 0.2 mg/day, or PARMODIA 0.4 mg/day was administered twice daily before or after breakfast and dinner for 52 weeks. The percent change in fasting serum TG at Week 24 and Week 52 (LOCF) was as presented in the following table.
Table 6. Percent change in fasting serum TG in Placebo/PARMODIA 0.2 mg/day group and PARMODIA groups (at Weeks 24 and 52):
| Treatment group and Baseline fasting serum TGa (mg/dL) | Percent change in fasting serum TGb | ||
|---|---|---|---|
| Time point | Percent change from baselinec (%) | Difference from placebo in percent changed (%) | |
| Placebo (up to Week 24) PARMODIA 0.2 mg/day (from Week 24) 284.3±117.6, n=57 | Week 24 | -10.814 [-17.933, -3.694] | - |
| Week 52 | -46.835 [-52.967, -40.704] | - | |
| PARMODIA 0.2 mg/day 240.3±93.5, n=54 | Week 24 | -44.347 [-51.656, -37.038] | -33.534 [-45.154,-21.914] |
| Week 52 | -43.629 [-49.924, -37.334] | - | |
| PARMODIA 0.4 mg/day | Week 24 | -45.093 [-52.283, -37.904] | -34.280 [-45.723,-22.836] |
| Week 52 | -46.552 [-52.744, -40.360] | - | |
a Mean ± SD To convert TG from mg/dL to mmol/L, multiply by 0.0113
b Analysis of covariance with baseline value as a covariate Last observation carried forward (LOCF) method was used.
c Least square mean [95% CI]
d Least square mean [Adjusted 95% CI]
5.2. Pharmacokinetic properties
Plasma pemafibrate concentration
(1) Single dose administration
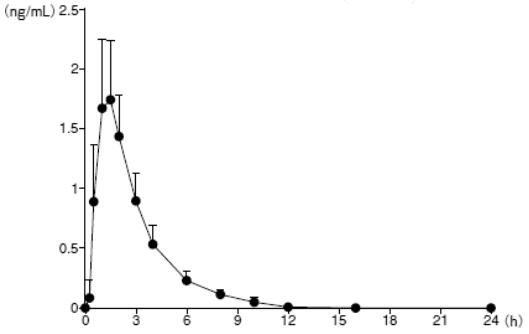
When a single dose of PARMODIA 0.1 mg was orally administered under fasted conditions to healthy Japanese adult males (16 subjects), the plasma concentration versus time and pharmacokinetic parameters are as presented in the following figure.
Figure. The plasma concentration versus time after a single oral dose in fasted healthy adult males:
Mean + SD (n=16)
Table 7. Pharmacokinetic parameters after a single oral dose in fasted healthy adult males:
| Cmax (ng/mL) | AUC0-inf (ng·h/mL) | tmax (h) | t1/2 (h) |
|---|---|---|---|
| 1.82±0.54 | 5.75±1.50 | 1.50 (1.00, 2.00) | 1.88±0.31 |
Cmax, AUC0-inf, t1/2: Mean ± SD
tmax: Median (Minimum, Maximum)
n=16
(2) Repeated dose administration
When PARMODIA 0.2 mg/day or 0.4 mg/day was orally administered twice daily after breakfast and dinner for 7 days to healthy Japanese adult males (8 subjects), the pharmacokinetic parameters on Day 1 and Day 7 are as presented in the following table. The plasma concentration reached a steady state on Day 2. The accumulation ratio based on AUC0-τ (repeated dosing/initial dosing, Mean ± SD) were 1.0997±0.0688 and 1.1169±0.1814, respectively.
Table 8. Pharmacokinetic parameters after repeated oral doses in healthy adult males:
| Dose of PARMODIA | Time point | Cmax (ng/mL) | AUC0-τ (ng·h/mL) | tmax (h) | t1/2 (h) |
|---|---|---|---|---|---|
| 0.2 mg/day Twice daily | Day 1 | 1.401±0.249 | 4.884±1.201 | 2.000 (1.00, 3.00) | - |
| Day 7 | 1.593±0.366 | 5.404±1.515 | 2.000 (1.00, 3.00) | 1.528±0.402 | |
| 0.4 mg/day Twice daily | Day 1 | 2.968±0.905 | 10.975±2.335 | 2.000 (1.00, 3.00) | - |
| Day 7 | 3.572±1.021 | 12.207±2.900 | 2.000 (1.00, 3.00) | 1.708±0.158 |
Cmax, AUC0-τ, t1/2: Mean ± SD, -: Not calculated
tmax: Median (Minimum, Maximum)
n=8
(3) Food effect
When a single dose of PARMODIA 0.1 mg was orally administered to healthy Japanese adult males (16 subjects), the ratio [90% CI] of geometric means of fasted state to fed state for Cmax and AUC0-t were 0.873 [0.803, 0.950] and 0.911 [0.863, 0.961].
Absorption
The absolute bioavailability of pemafibrate was 61.5% (Data for non-Japanese subjects).
Plasma protein binding ratio
The human plasma protein binding ratio of pemafibrate was ≥99%.
Metabolism
(1) When a single dose of 14C-pemafibrate was orally administered to healthy adult subjects, the main metabolites in plasma were an oxidized form at the benzyl position, and a mixture of glucuronide conjugate of dicarboxylated form and N-dealkylated form (Data for non-Japanese subjects).
(2) Pemafibrate is a substrate of CYP2C8, CYP2C9, CYP3A4, CYP3A7, UGT1A1, UGT1A3, and UGT1A8 (in vitro).
Excretion
(1) When a single dose of 14C-pemafibrate was administered to healthy adult subjects, excretion of radioactivity in urine and feces up to 216 hours after administration was 14.53% and 73.29%, respectively (Data for non-Japanese subjects). Pemafibrate is excreted mainly in the feces.
(2) Pemafibrate is a substrate of P-gp, BCRP, OATP1A2, OATP1B1, OATP1B3, OCT2, and NTCP (in vitro).
Drug interactions
(1) Co-administration with cyclosporin, rifampicin, clopidogrel, clarithromycin, fluconazole, digoxin, or warfarin
When PARMODIA was co-administered with each drug in healthy adult subjects (non-Japanese), the effect on the pharmacokinetic parameters was as presented in the following table.
Table 9. Effect of co-administration of PARMODIA and each drug on pharmacokinetic parameters (data for non-Japanese subjects):
| Co-administrated drug | Dose of Co- administrated drug | Dose of PARMODIA | Analyte | Ratio of geometric means [90% CI] (Combination therapy/monotherapy) | |
|---|---|---|---|---|---|
| Cmax | AUC0-inf | ||||
| Cyclosporine | 600 mg Single-dose | 0.4 mg Single-dose | PARMODIA | 8.9644 [7.5151, 10.6931] n=14 | 13.9947 [12.6175,15.5223] n=12 |
| Rifampicin | 600 mg Single-dose | 0.4 mg Single-dose | PARMODIA | 9.4336 [8.3626, 10.6419] n=20 | 10.9009 [9.9154, 11.9844] n=17 |
| 600 mg/day Once daily 10 days Monotherapy | 0.4 mg Monotherapy | PARMODIA | 0.3792a [0.3378, 0.4257] n=20 | 0.2221a [0.2065, 0.2389] n=16 | |
| Clopidogrel | 300 mg Single dose Day 4 | 0.4 mg Single dose Day 4 | PARMODIA | 1.4855 [1.3915, 1.5858] n=20 | 2.3728 [2.2473, 2.5052] n=20 |
| 75 mg/day Once daily 5 days Days 5 to 9 | 0.4 mg Single-dose Day 7 | PARMODIA | 1.3415 [1.2583, 1.4302] n=20 | 2.0876 [1.9811, 2.1998] n=20 | |
| Clarithromycin | 1,000 mg/day Twice daily 8 days | 0.4 mg Single-dose | PARMODIA | 2.4246 [2.1632, 2.7174] n=18 | 2.0975 [1.9158, 2.2964] n=17 |
| Fluconazole | 400 mg/day Once daily 11 days | 0.4 mg Single-dose | PARMODIA | 1.4409 [1.2899, 1.6096] n=19 | 1.7891 [1.6638, 1.9239] n=17 |
| Digoxin | 0.5 mg/day Twice daily (Day 1), 0.25 mg/day Once daily 16 days | 0.8 mg/day Twice daily 6 days Days 11 to 16 | Digoxin | 1.0325 [0.9511, 1.1210] n=19 | 0.9463b [0.9090, 0.9850] n=19 |
| Warfarin* | 5 mg/day Once daily (Day 1 and Day 2), Maintenance dosec Once daily 21 days | 0.4 mg/day Twice daily 8 days Days 14 to 21 | R-warfarin | 1.004 [0.972, 1.037] n=19 | 1.029b [1.004, 1.055] n=19 |
| S-warfarin | 0.929 [0.889, 0.970] n=19 | 0.951b [0.926, 0.976] n=19 | |||
a Geometric mean ratios [90% CI] of PARMODIA monotherapy after repeated administration of rifampicin to PARMODIA monotherapy before repeated administration of rifampicin for Cmax and AUC0-inf.
b AUC0-τ
c On Day 3 through Day 9, the dosage was adjusted to achieve an international normalized ratio of prothrombin time (PT-INR) of 1.2 to 2.2. On Day 10 and thereafter, the maintenance dose that achieved PT-INR of 1.2 to 2.2 was administered.
* Least square mean ratios [90% CI] of repeated co-administration of warfarin with PARMODIA to repeated warfarin monotherapy for PT-INR and PT were 1.0196 [0.9878, 1.0514] (n=19) and 1.0191 [0.9869, 1.0512] (n=19).
Note: The approved dosage and administration of PARMODIA is an oral dose of 0.1 mg twice daily, and the maximum dosage is an oral dose of 0.2 mg twice daily (see section 4.2).
(2) Co-administration with HMG-CoA reductase inhibitors
When PARMODIA and HMG-CoA reductase inhibitors were co-administered to healthy adult males (Japanese and non-Japanese), the effect of coadministration on the pharmacokinetic parameters was as presented in the following table.
Table 10. Effect of co-administration of PARMODIA and each drug on pharmacokinetic parameters (data for Japanese and non-Japanese subjects):
| Co-administrated drug | Dose of co- administrated drug | Dose of PARMODIA | Analyte | Ratio of geometric means [90% CI] (Combination therapy/monotherapy) | |
|---|---|---|---|---|---|
| Cmax | AUC0-τ | ||||
| Atorvastatin | 20 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (n=18) | 1.166 [1.069, 1.272] | 1.098 [1.016, 1.187] |
| Atorvastatin (n=18) | 1.032 [0.960, 1.109] | 0.934 [0.851, 1.024] | |||
| o-hydroxyatorvastatin | 0.875 [0.826, 0.927] | 0.784 [0.736, 0.836] | |||
| Simvastatin | 20 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (n=18) | 1.230 [1.090, 1.388] | 1.125 [0.997, 1.270] |
| Simvastatin (n=19) | 0.858 [0.660, 1.114] | 0.846 [0.722, 0.992] | |||
| Open acid form of simvastatin (n=19) | 0.626 [0.541, 0.725] | 0.405 [0.345, 0.475] | |||
| Pitavastatin | 4 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (n=18) | 1.061 [0.970, 1.160] | 1.122 [1.041, 1.209] |
| Pitavastatin (n=18) | 1.011 [0.973, 1.050] | 1.036 [1.007, 1.066] | |||
| Pravastatin | 20 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (n=18) | 1.058 [0.964, 1.162] | 1.057 [1.013, 1.102] |
| Pravastatin (n=18) | 1.107 [0.908, 1.351] | 1.065 [0.922, 1.231] | |||
| Fluvastatin | 60 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (n=18) | 1.181 [1.080, 1.290] | 1.207 [1.144, 1.274] |
| Fluvastatin (n=18) | 0.989 [0.790, 1.239] | 1.151 [1.057, 1.253] | |||
| Rosuvastatin | 20 mg/day Once daily 7 days | 0.4 mg/day Twice daily 7 days | PARMODIA (non-Japanese subjects, n=24) | 1.106 [1.048, 1.167] | 1.110 [1.046, 1.177] |
| Rosuvastatin (non-Japanese subjects, n=24) | 1.092 [1.016, 1.174] | 1.025 [0.964, 1.091] | |||
Special populations
Pharmacokinetics in Patients with Fatty Liver and Patients with Hepatic Cirrhosis
When a single dose of PARMODIA 0.2 mg was orally administered to Japanese patients with fatty liver and patients with hepatic cirrhosis, the ratios of pharmacokinetic parameters (patients with fatty liver or with hepatic cirrhosis to subjects with normal hepatic function) were as presented in the following table. Compared with subjects with normal hepatic function, the exposure was higher in patients with fatty liver and patients with hepatic cirrhosis.
Table 11. Ratios [90% CI] of geometric means of patients with fatty liver or hepatic cirrhosis to subjects with normal hepatic function (n=8) for Cmax and AUC0-t:
| Cmax | AUC0-t | |
|---|---|---|
| Fatty liver group (n=10) | 1.198 [0.819, 1.750] | 1.194 [0.836, 1.707] |
| Mild hepatic cirrhosis Child-Pugh grade A group (n=8) | 2.329 [1.561, 3.475] | 2.076 [1.425, 3.026] |
| Moderate hepatic cirrhosis Child-Pugh grade B group (n=6) | 3.882 [2.520, 5.980] | 4.191 [2.790, 6.294] |
Pharmacokinetics in Patients with Renal Impairment
When a single dose of PARMODIA 0.2 mg was orally administered to Japanese patients with renal impairment (mild, moderate, severe, or end-stage renal failure), the ratios of pharmacokinetic parameters (patients with renal impairment to subjects with normal renal function) were as presented in the following table. Compared with subjects with normal renal function, the exposure was higher in patients with renal impairment; however, the exposure did not increase as the renal function reduced.
Table 12. Ratios [90% CI] of geometric means of patients with renal impairment to subjects with normal renal function (n=8) for Cmax and AUC0-t:
| Cmax | AUC0-t | |
|---|---|---|
| Mild renal impairment group [50 ≤ Ccr < 80 mL/min] (n=8) | 1.644 [1.155, 2.342] | 1.629 [1.161, 2.287] |
| Moderate renal impairment group [30 ≤ Ccr < 50 mL/min] (n=8) | 1.093 [0.767, 1.556] | 1.154 [0.822, 1.620] |
| Severe renal impairment group [Ccr < 30 mL/min] (n=7) | 1.545 [1.072, 2.228] | 1.296 [0.913, 1.841] |
| End-stage renal failure group [Undergoing hemodialysis] (n=7) | 1.258 [0.872, 1.813] | 1.607 [1.131, 2.282] |
PARMODIA was orally administered at a dose of 0.1 mg twice daily in morning and evening for 12 weeks to patients with dyslipidemia accompanied by high TG value and renal impairment (severe renal impairment with eGFR <30 mL/min/1.73 m² or on dialysis and mild-to-moderate renal impairment with eGFR ≥30 and <60 mL/min/1.73 m²). The ratio and 90% CI of geometric means of AUC0-τ in patients with severe renal impairment compared to those with mild-to-moderate renal impairment (control group) at Week 12 were as presented in the following table. The level of exposure did not increase in patients with severe renal impairment.
Table 13. Ratio [90% CI] of geometric means of AUC0-τ in patients with severe renal impairment (n=8) compared to those with mild-tomoderate renal impairment (n=7):
| Ratio of geometric means of AUC0-τ [90% CI] | |
|---|---|
| Severe renal impairment group [eGFR <30 mL/min/1.73 m² or on dialysis] | 0.9177 [0.6198,1.3587] |
Pharmacokinetic parameters were presented in the following table.
Table 14. Pharmacokinetic parameters after repeated oral administration in patients with dyslipidemia accompanied by high TG value and renal impairment:
| Cmax (ng/mL) | AUC0-τ (ng ∙ h/mL) | |
|---|---|---|
| Mild-to-moderate renal impairment group [30≤ eGFR <60 mL/min/1.73 m²] (n=7) | 2.4483±0.9535 | 8.6994±4.0397 |
| Severe renal impairment group [eGFR <30 mL/min/1.73 m²] (n=4) | 2.0508±0.6588 | 7.4130±3.9548 |
| Severe renal impairment group [dialysis] (n=4) | 1.8798±0.5728 | 8.4470±3.3054 |
Mean ± SD
5.3. Preclinical safety data
In a carcinogenicity study in mice (≥0.075 mg/kg/day), an increase in the incidence of hepatocellular carcinomas and hepatocellular adenomas was observed. In a carcinogenicity study in rats (≥0.3 mg/kg/day in male rats and ≥1 mg/kg/day in female rats), an increase in the incidence of hepatocellular carcinomas, hepatocellular adenomas, pancreatic acinar cell carcinomas, pancreatic acinar cell adenomas, testicular Leydig cell adenomas, and thyroidal follicular epithelial cell adenomas was observed. All of these findings are considered to be specific to rodents.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.