TRESIBA Solution for injection Ref.[107042] Active ingredients: Insulin degludec
Source: FDA, National Drug Code (US) Revision Year: 2023
12.1. Mechanism of Action
The primary activity of insulin, including TRESIBA, is regulation of glucose metabolism. Insulin and its analogs lower blood glucose by stimulating peripheral glucose uptake, especially by skeletal muscle and fat, and by inhibiting hepatic glucose production. Insulin also inhibits lipolysis and proteolysis, and enhances protein synthesis. TRESIBA forms multi-hexamers when injected into the subcutaneous tissue resulting in a subcutaneous insulin degludec depot. The protracted time action profile of TRESIBA is predominantly due to delayed absorption of insulin degludec from the subcutaneous tissue to the systemic circulation and to a lesser extent due to binding of insulin degludec to circulating albumin.
12.2. Pharmacodynamics
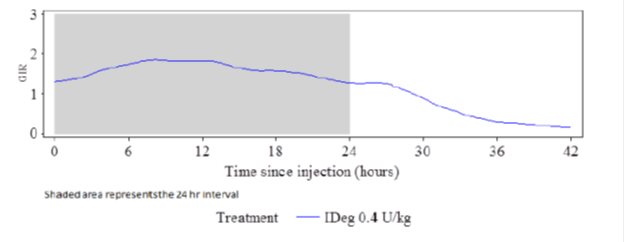
The glucose-lowering effect of TRESIBA after 8 days of once-daily dosing was measured in a euglycemic glucose clamp study enrolling 21 patients with type 1 diabetes. Figure 2 shows the pharmacodynamic effect of TRESIBA over time following 8 once-daily subcutaneous injections of 0.4 U/kg of TRESIBA in patients with type 1 diabetes.
Figure 2. Mean GIR Profile for 0.4 units/kg Dose of TRESIBA (Steady State) in Patients with Type 1 Diabetes Mellitus:
The mean maximum glucose lowering effect (GIRmax) of a 0.4 units/kg dose of TRESIBA was 2.0 mg/kg/min, which was observed at a median of 12 hours post-dose. The glucose lowering effect of TRESIBA lasted at least 42 hours after the last of 8 once-daily injections.
In patients with type 1 diabetes mellitus, the steady-state, within subjects, day-to-day variability in total glucose lowering effect was 20% with TRESIBA (within-subject coefficient of variation for AUCGIR,τ,SS).
The total glucose-lowering effect of TRESIBA over 24 hours measured in a euglycemic clamp study after 8 days of once-daily administration in patients with type 1 diabetes increases approximately in proportion to the dose for doses between 0.4 units/kg to 0.8 units/kg.
The total glucose-lowering effect of 0.4 units/kg of TRESIBA U-100 and 0.4 units/kg of TRESIBA U-200, administered at the same dose, and assessed over 24 hours in a euglycemic clamp study after 8 days of once-daily injection was comparable.
12.3. Pharmacokinetics
Absorption
In patients with type 1 diabetes, after 8 days of once daily subcutaneous dosing with 0.4 units/kg of TRESIBA, maximum insulin degludec concentrations of 4472 pmol/L were attained at a median of 9 hours (tmax). After the first dose of TRESIBA, median onset of appearance was around one hour.
Total insulin degludec concentration (i.e., exposure) increased in a dose proportional manner after subcutaneous administration of 0.4 units/kg to 0.8 units/kg TRESIBA. Total and maximum insulin degludec exposure at steady state are comparable between TRESIBA U-100 and TRESIBA U-200 when each is administered at the same units/kg dose.
Insulin degludec concentration reached steady state levels after 3-4 days of TRESIBA administration [see Dosage and Administration (2.2)].
Distribution
The affinity of insulin degludec to serum albumin corresponds to a plasma protein binding of >99% in human plasma. The results of the in vitro protein binding studies demonstrate that there is no clinically relevant interaction between insulin degludec and other protein bound drugs.
Elimination
The half-life after subcutaneous administration is determined primarily by the rate of absorption from the subcutaneous tissue. On average, the half-life at steady state is approximately 25 hours independent of dose. Degradation of TRESIBA is similar to that of insulin human; all metabolites formed are inactive. The mean apparent clearance of insulin degludec is 0.03 L/kg (2.1 L/h in 70 kg patient) after single subcutaneous dose of 0.4 units/kg.
Specific Populations
Pediatrics
Population pharmacokinetic analysis was conducted for TRESIBA using data from 199 pediatric subjects (1 to <18 years of age) with type 1 diabetes. Body weight was a significant covariate affecting the clearance of TRESIBA. After adjusting for body weight, the total exposure of TRESIBA at steady state was independent of age.
Geriatrics
Pharmacokinetic and pharmacodynamic response of TRESIBA was compared in 13 younger adult (18−35 years) and 14 geriatric (≥65 years) subjects with type 1 diabetes following two 6-day periods of once-daily subcutaneous dosing with 0.4 units/kg dose of TRESIBA or insulin glargine. On average, the pharmacokinetic and pharmacodynamic properties of TRESIBA at steady-state were similar in younger adult and geriatric subjects, albeit with greater between subject variability among the geriatric subjects.
Gender
The effect of gender on the pharmacokinetics of TRESIBA was examined in an across-trial analysis of the pharmacokinetic and pharmacodynamic studies conducted using unit/kg doses of TRESIBA. Overall, there were no clinically relevant differences in the pharmacokinetic properties of insulin degludec between female and male subjects.
Obesity
The effect of BMI on the pharmacokinetics of TRESIBA was explored in a cross-trial analysis of pharmacokinetic and pharmacodynamic studies conducted using unit/kg doses of TRESIBA. For subjects with type 1 diabetes, no relationship between exposure of TRESIBA and BMI was observed. For subjects with type 1 and type 2 diabetes a trend for decrease in glucose-lowering effect of TRESIBA with increasing BMI was observed.
Race and Ethnicity
TRESIBA has been studied in a pharmacokinetic and pharmacodynamic study in Black or African American subjects not of Hispanic or Latino origin (N=18), White subjects of Hispanic or Latino origin (N=22) and White subjects not of Hispanic or Latino origin (N=23) with type 2 diabetes mellitus conducted using unit/kg doses of TRESIBA. There were no statistically significant differences in the pharmacokinetic and pharmacodynamic properties of TRESIBA between the racial and ethnic groups investigated.
Pregnancy
The effect of pregnancy on the pharmacokinetics and pharmacodynamics of TRESIBA has not been studied [see Use in Specific Populations (8.1)].
Renal Impairment
TRESIBA pharmacokinetics was studied in 32 subjects (N=4-8/group) with normal or impaired renal function/end-stage renal disease following administration of a single subcutaneous dose (0.4 units/kg) of TRESIBA. Renal function was defined using creatinine clearance (Clcr) as follows: ≥90 mL/min (normal), 60-89 mL/min (mild), 30-59 mL/min (moderate) and <30 mL/min (severe). Subjects requiring dialysis were classified as having end-stage renal disease (ESRD). Total (AUCIDeg,0-120h,SD) and peak exposure of TRESIBA were on average about 10-25% and 13-27% higher, respectively in subjects with mild to severe renal impairment except subjects with ESRD who showed similar exposure as compared to subjects with normal renal function. No systematic trend was noted for this increase in exposure across different renal impairment subgroups. Hemodialysis did not affect clearance of TRESIBA (CL/FIDeg,SD) in subjects with ESRD [see Use in Specific Populations (8.6)].
Hepatic Impairment
TRESIBA has been studied in a pharmacokinetic study in 24 subjects (N=6/group) with normal or impaired hepatic function (mild, moderate, and severe hepatic impairment) following administration of a single subcutaneous dose (0.4 units/kg) of TRESIBA. Hepatic function was defined using Child-Pugh Scores ranging from 5 (mild hepatic impairment) to 15 (severe hepatic impairment). No differences in the pharmacokinetics of TRESIBA were identified between healthy subjects and subjects with hepatic impairment [see Use in Specific Populations (8.7)].
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Standard 2-year carcinogenicity studies in animals have not been performed to evaluate the carcinogenic potential of insulin degludec. In a 52-week study including human insulin (NPH insulin) as comparator (6.7 units/kg/day), Sprague-Dawley rats were dosed subcutaneously with insulin degludec at 3.3, 6.7, and 10 units/kg/day, resulting in 5 times the human exposure (AUC) when compared to a human subcutaneous dosage of 0.75 units/kg/day. Human insulin was dosed at 6.7 units/kg/day. No treatment-related increases in incidences of hyperplasia, benign or malignant tumors were recorded in female mammary glands from rats dosed with insulin degludec and no treatment related changes in the female mammary gland cell proliferation were found using BrdU incorporation. Further, no treatment related changes in the occurrence of hyperplastic or neoplastic lesions were seen in other tissues in animals dosed with insulin degludec when compared to vehicle or human insulin.
Genotoxicity testing of insulin degludec was not performed.
In a combined fertility and embryo-fetal study in male and female rats, treatment with insulin degludec up to 21 units/kg/day (approximately 5 times the human subcutaneous dose of 0.75 units/kg/day, based on units/body surface area) prior to mating and in female rats during gestation had no effect on mating performance and fertility.
14. Clinical Studies
The efficacy of TRESIBA administered once-daily either at the same time each day or at any time each day in patients with type 1 diabetes and used in combination with a mealtime insulin was evaluated in three randomized, open-label, treat-to-target, active-controlled trials in adults and one randomized, open-label, treat-to-target, active-controlled trial in pediatric patients 1 year of age and older. The efficacy of TRESIBA administered once-daily either at the same time each day or at any time each day in adult patients with type 2 diabetes and used in combination with a mealtime insulin or in combination with common oral anti-diabetic agents was evaluated in six randomized, open-label, treat-to-target active-controlled trials.
Adult patients treated with TRESIBA achieved levels of glycemic control similar to those achieved with LANTUS (insulin glargine 100 units/mL) and LEVEMIR (insulin detemir) and achieved statistically significant improvements compared to sitagliptin.
14.1 Type 1 Diabetes – Adult
TRESIBA Administered at the Same Time Each Day in Combination with a Rapid-Acting Insulin Analog at Mealtimes in Adult Patients
Study A:
The efficacy of TRESIBA was evaluated in a 52-week randomized, open-label, multicenter trial in 629 patients with type 1 diabetes mellitus (Study A). Patients were randomized to TRESIBA once-daily with the evening meal or insulin glargine U-100 once-daily according to the approved labeling. Insulin aspart was administered before each meal in both treatment arms.
The mean age of the trial population was 43 years and mean duration of diabetes was 19 years. 59% were male. 93% were White, 2% Black or African American. 5% were Hispanic. 9% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 26.3 kg/m².
At week 52, the difference in HbA1c reduction from baseline between TRESIBA and insulin glargine U-100 was -0.01% with a 95% confidence interval of [-0.14%; 0.11%] and met the pre-specified non-inferiority margin (0.4%). See Table 6, Study A.
Study B:
The efficacy of TRESIBA was evaluated in a 26-week randomized, open-label, multicenter trial in 455 patients with type 1 diabetes mellitus (Study B). Patients were randomized to TRESIBA or insulin detemir once-daily in the evening. After 8 weeks, insulin detemir could be dosed twice-daily. 67% used insulin detemir once daily at end of trial. 33% used insulin detemir twice daily at end of trial. Insulin aspart was administered before each meal in both treatment arms.
The mean age of the trial population was 41 years and mean duration of diabetes was 14 years. 52% were male. 45% were White, 0.4% Black or African American. 4% were Hispanic. 4% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 23.9 kg/m².
At week 26, the difference in HbA1c reduction from baseline between TRESIBA and insulin detemir was -0.09% with a 95% confidence interval of [-0.23%; 0.05%] and met the pre-specified non-inferiority margin (0.4%). See Table 6, Study B.
Table 6. Results at Week 52 in a Trial Comparing TRESIBA to Insulin Glargine U-100 (Study A) and Week 26 in a Trial Comparing TRESIBA to Insulin Detemir (Study B) in Adult Patients with Type 1 Diabetes Mellitus Receiving Insulin Aspart at Mealtimes:
| Study A | Study B | |||
|---|---|---|---|---|
| TRESIBA + Insulin aspart | Insulin glargine U-100 + Insulin aspart | TRESIBA + Insulin aspart | Insulin detemir + Insulin aspart | |
| N | 472 | 157 | 302 | 153 |
| HbA1c (%) | ||||
| Baseline | 7.7 | 7.7 | 8.0 | 8.0 |
| End of trial | 7.3 | 7.3 | 7.3 | 7.3 |
| Adjusted mean change from baseline* | -0.36 | -0.34 | -0.71 | -0.61 |
| Estimated treatment difference [95%CI] TRESIBA - basal insulin U-100 | -0.01 [-0.14;0.11] | -0.09 [-0.23;0.05] | ||
| Proportion Achieving HbA1c <7% at Trial End | 39.8% | 42.7% | 41.1% | 37.3% |
| FPG (mg/dL) | ||||
| Baseline | 165 | 174 | 178 | 171 |
| End of trial | 141 | 149 | 131 | 161 |
| Adjusted mean change from baseline | -27.6 | -21.6 | -43.3 | -13.5 |
| Daily basal insulin dose | ||||
| Baseline mean | 28 U | 26 U | 22 U | 22 U |
| Mean dose at end of study | 29 U1 | 31 U1 | 25 U2 | 29 U2 |
| Daily bolus insulin dose | ||||
| Baseline mean | 29 U | 29 U | 28 U | 31 U |
| Mean dose at end of study | 32 U1 | 35 U1 | 36 U2 | 41 U2 |
1 At Week 52
2 At Week 26
* The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study A, there were 14.8% of subjects in the TRESIBA and 11.5% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
In Study B, there were 6.3% of subjects in the TRESIBA and 9.8% Insulin detemir arms for whom data was missing at the time of the HbA1c measurement.
Study C: TRESIBA Administered at the Same Time Each Day or at Any Time Each Day in Combination with a Rapid-Acting Insulin Analog at Mealtimes in Adult Patients:
The efficacy of TRESIBA was evaluated in a 26-week randomized, open-label, multicenter trial in 493 patients with type 1 diabetes mellitus. Patients were randomized to TRESIBA injected once-daily at the same time each day (with the main evening meal), to TRESIBA injected once daily at any time each day or to insulin glargine U-100 injected once-daily according to the approved labeling. The any time each day TRESIBA arm was designed to simulate a worst-case scenario injection schedule of alternating short and long, once daily, dosing intervals (i.e., alternating intervals of 8 to 40 hours between doses). TRESIBA in this arm was dosed in the morning on Monday, Wednesday, and Friday and in the evening on Tuesday, Thursday, Saturday, and Sunday. Insulin aspart was administered before each meal in all treatment arms.
The mean age of the trial population was 43.7 years and mean duration of diabetes was 19 years. 58% were male. 98% were White, 2% Black or African American. 3% were Hispanic. 7% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 26.7 kg/m².
At week 26, the difference in HbA1c reduction from baseline between TRESIBA administered at alternating times and insulin glargine U-100 was 0.17% with a 95% confidence interval of [0.04%; 0.30%] and met the pre-specified non-inferiority margin (0.4%). See Table 7.
Table 7. Results at Week 26 in a Trial Comparing TRESIBA Dosed Once Daily at the Same and at Alternating Times Each Day to Insulin Glargine U-100 in Adult Patients with Type 1 Diabetes Mellitus Receiving Insulin Aspart at Mealtimes
| TRESIBA at the same time each day + Insulin aspart | TRESIBA at alternating times + Insulin aspart | Insulin glargine U-100 + Insulin aspart | |
|---|---|---|---|
| N | 165 | 164 | 164 |
| HbA1c (%) | |||
| Baseline | 7.7 | 7.7 | 7.7 |
| End of trial | 7.3 | 7.3 | 7.1 |
| Adjusted mean change from baseline * | -0.41 | -0.40 | -0.57 |
| Estimated treatment difference [95%CI] TRESIBA alternating - Insulin glargine U-100 | 0.17 [0.04;0.30] | ||
| Proportion Achieving HbA1c <7% at Trial End | 37.0% | 37.2% | 40.9% |
| FPG (mg/dL) | |||
| Baseline | 179 | 173 | 175 |
| End of trial | 133 | 149 | 151 |
| Adjusted mean change from baseline | -41.8 | -24.7 | -23.9 |
| Daily basal insulin dose | |||
| Baseline mean | 28 U | 29 U | 29 U |
| Mean dose at end of study | 32 U | 36 U | 35 U |
| Daily bolus insulin dose | |||
| Baseline mean | 29 U | 33 U | 32 U |
| Mean dose at end of study | 27 U | 30 U | 35 U |
* The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study C, there were 15.8% and 15.9% of subjects in the TRESIBA (same time and alternating times respectively) and 7.9% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
14.2 Type 1 Diabetes – Pediatric Patients 1 Year of Age and Older
Study J: TRESIBA Administered at the Same Time Each Day in Combination with a Rapid-Acting Insulin Analog at Mealtimes in Pediatric Patients 1 Year of Age and Older
The efficacy of TRESIBA was evaluated in a 26-week, randomized, open label, multicenter trial in 350 patients with type 1 diabetes mellitus (Study J). Patients were randomized to TRESIBA once-daily or insulin detemir once or twice-daily. Subjects on a twice-daily insulin detemir regimen were dosed at breakfast and in the evening either with the main evening meal or at bedtime. Insulin aspart was administered before each main meal in both treatment arms. At end of trial, 36% used insulin detemir once daily and 64% used insulin detemir twice daily.
The mean age of the trial population was 10 years; 24% were ages 1-5 years; 39% were ages 6-11 years and 36% were ages 12-17 years. The mean duration of diabetes was 4 years. 55% were male. 75% were White, 3% Black or African American. 3% were Hispanic. The mean z-score for body weight was 0.31.
At week 26, the difference in HbA1c reduction from baseline between TRESIBA and insulin detemir was 0.15% with a 95% confidence interval of [-0.03%; 0.33%] and met the pre-specified non-inferiority margin (0.4%). See Table 8.
Table 8. Results at Week 26 in a Trial Comparing TRESIBA to Insulin Detemir in Pediatric Patients 1 Year of Age and Older with Type 1 Diabetes Mellitus Receiving Insulin Aspart at Mealtimes:
| TRESIBA + Insulin aspart | Insulin detemir + Insulin aspart | |
|---|---|---|
| N | 174 | 176 |
| HbA1c (%) | ||
| Baseline | 8.2 | 8.0 |
| End of 26 weeks | 8.0 | 7.7 |
| Adjusted mean change from baseline after 26 weeks± | -0.19 | -0.34 |
| Estimated treatment difference [95%CI] TRESIBA v. Insulin detemir | 0.15 [ -0.03; 0.33] | |
| FPG (mg/dL) | ||
| Baseline | 162 | 151 |
| End of 26 weeks | 150 | 160 |
| Adjusted mean change from baseline after 26 weeks | 52.0 | 59.6 |
| Daily basal insulin dose | ||
| Baseline mean | 15 U (0.37 U/kg) | 16 U (0.41 U/kg) |
| Mean dose after 26 weeks | 16 U (0.37 U/kg) | 22 U (0.51 U/kg) |
| Daily bolus insulin dose | ||
| Baseline mean | 20 U (0.50 U/kg) | 20 U (0.52 U/kg) |
| Mean dose after 26 weeks | 23 U (0.56 U/kg) | 22 U (0.57 U/kg) |
± The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with missing data imputed by multiple imputation carrying forward the baseline value and adding the error term, with treatment, region, sex, and age group as fixed factors, and baseline HbA1c as covariate.
In Study J, there were 2.9% of subjects in TRESIBA and 6.3% Insulin detemir arms for whom data was missing at the 26-week HbA1c measurement.
14.3 Type 2 Diabetes - Adult
Study D: TRESIBA Administered at the Same Time Each Day as an Add-on to Metformin with or without a DPP-4 Inhibitor in Insulin Naïve Adult Patients
The efficacy of TRESIBA was evaluated in a 52-week randomized, open-label, multicenter trial that enrolled 1030 insulin naïve patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agents (OADs). Patients were randomized to TRESIBA once-daily with the evening meal or insulin glargine U-100 once-daily according to the approved labeling. Metformin alone (83%) or in combination with a DPP-4 inhibitor (18%) was used as background therapy in both treatment arms.
The mean age of the trial population was 59 years and mean duration of diabetes was 9 years. 62% were male. 88% were White, 7% Black or African American. 17% were Hispanic. 10% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 31.1 kg/m².
At week 52, the difference in HbA1c reduction from baseline between TRESIBA and insulin glargine U-100 was 0.09% with a 95% confidence interval of [-0.04%; 0.22%] and met the pre-specified non-inferiority margin (0.4%); See Table 9.
Table 9. Results at Week 52 in a Trial Comparing TRESIBA to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus on OAD (s)*:
| TRESIBA + OAD (s)* | Insulin glargine U-100 + OAD (s)* | |
|---|---|---|
| N | 773 | 257 |
| HbA1c (%) | ||
| Baseline | 8.2 | 8.2 |
| End of trial | 7.1 | 7.0 |
| Adjusted mean change from baseline** | -1.06 | -1.15 |
| Estimated treatment difference [95%CI] TRESIBA - Insulin glargine U-100 | 0.09 [-0.04;0.22] | |
| Proportion Achieving HbA1c <7% at Trial End | 51.7% | 54.1% |
| FPG (mg/dL) | ||
| Baseline | 174 | 174 |
| End of trial | 106 | 115 |
| Adjusted mean change from baseline | -68.0 | -60.2 |
| Daily insulin dose | ||
| Baseline mean (starting dose) | 10 U | 10 U |
| Mean dose after 52 weeks | 56 U | 58 U |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study D, there were 20.6% of subjects in the TRESIBA and 22.2% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
Study E: TRESIBA U-200 Administered at the Same Time Each Day as an Add-on to Metformin with or without a DPP-4 Inhibitor in Insulin Naïve Adult Patients
The efficacy of TRESIBA U-200 was evaluated in a 26-week randomized, open-label, multicenter trial in 457 insulin naïve patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agents (OADs) at baseline. Patients were randomized to TRESIBA U-200 once-daily with the evening meal or insulin glargine U-100 once-daily according to the approved labeling. Both treatment arms were receiving metformin alone (84%) or in combination with a DPP-4 inhibitor (16%) as background therapy.
The mean age of the trial population was 58 years and mean duration of diabetes was 8 years. 53% were male. 78% were White, 14% Black or African American. 8% were Hispanic. 8% of patients had eGFR <60 mL/min/1.73m². The mean BMI was approximately 32.4 kg/m².
At week 26, the difference in HbA1c reduction from baseline between TRESIBA U-200 and insulin glargine U-100 was 0.04% with a 95% confidence interval of [-0.11%; 0.19%] and met the pre-specified non-inferiority margin (0.4%). See Table 10.
Table 10. Results at Week 26 in a Trial Comparing TRESIBA U-200 to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus on OAD (s)*:
| TRESIBA U-200 + Met ± DPP-4 | Insulin glargine U-100 + Met ± DPP-4 | |
|---|---|---|
| N | 228 | 229 |
| HbA1c (%) | ||
| Baseline | 8.3 | 8.2 |
| End of trial | 7.0 | 6.9 |
| Adjusted mean change from baseline** | -1.18 | -1.22 |
| Estimated treatment difference [95%CI] TRESIBA - Insulin glargine U-100 | 0.04 [-0.11;0.19] | |
| Proportion Achieving HbA1c <7% at Trial End | 52.2% | 55.9% |
| FPG (mg/dL) | ||
| Baseline | 172 | 174 |
| End of trial | 106 | 113 |
| Adjusted mean change from baseline | -71.1 | -63.5 |
| Daily insulin dose | ||
| Baseline mean | 10 U | 10 U |
| Mean dose after 26 weeks | 59 U | 62 U |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study E, there were 12.3% of subjects in the TRESIBA and 12.7% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
Study F: TRESIBA Administered at the Same Time Each Day in Insulin Naïve Adult Patients as an Add-on to One or More of the Following Oral Agents: Metformin, Sulfonylurea, Glinides or Alpha-Glucosidase Inhibitors
The efficacy of TRESIBA was evaluated in a 26-week randomized, open-label, multicenter trial in Asia in 435 insulin naïve patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agents (OADs) at baseline. Patients were randomized to TRESIBA once-daily in the evening or insulin glargine U-100 once-daily according to the approved labeling. Pre-trial oral antidiabetes agents were continued as background therapy except for DPP-4 inhibitors or thiazolidinediones in both treatment arms.
The mean age of the trial population was 59 years and mean duration of diabetes was 12 years. 54% were male. All patients were Asian. 11% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 25.0 kg/m².
At week 26, the difference in HbA1c reduction from baseline between TRESIBA and insulin glargine U-100 was 0.11% with a 95% confidence interval of [-0.03%; 0.24%] and met the pre-specified non-inferiority margin (0.4%). See Table 11.
Table 11. Results at Week 26 in a Trial Comparing TRESIBA to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus on OAD (s)*:
| TRESIBA + OAD (s)* | Insulin glargine U-100 + OAD (s)* | |
|---|---|---|
| N | 289 | 146 |
| HbA1c (%) | ||
| Baseline | 8.4 | 8.5 |
| End of trial | 7.2 | 7.1 |
| Adjusted mean change from baseline** | -1.42 | -1.52 |
| Estimated treatment difference [95%CI] TRESIBA - Insulin glargine U-100 | 0.11 [-0.03 ; 0.24] | |
| Proportion Achieving HbA1c <7% at Trial End | 40.8% | 48.6% |
| FPG (mg/dL) | ||
| Baseline | 152 | 156 |
| End of trial | 100 | 102 |
| Adjusted mean change from baseline | -54.6 | -53.0 |
| Daily insulin dose | ||
| Baseline mean (starting dose) | 9 U | 9 U |
| Mean dose after 26 weeks | 19 U | 24 U |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study F, there were 10% of subjects in the TRESIBA and 6.8% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
Study G: TRESIBA Administered at the Same Time Each Day or Any Time Each Day as an Add-on to One and up to Three of the Following Oral Agents: Metformin, Sulfonylurea or Glinides or Pioglitazone in Adult Patients
The efficacy of TRESIBA was evaluated in a 26-week randomized, open-label, multicenter trial in 687 patients with type 2 diabetes mellitus inadequately controlled on basal insulin alone, oral antidiabetic agents (OADs) alone or both basal insulin and OAD. Patients were randomized to TRESIBA injected once-daily at the same time each day (with the main evening meal), to TRESIBA injected once daily at any time each day or to insulin glargine U-100 injected once-daily according to the approved labeling. The any time each day TRESIBA arm was designed to simulate a worst-case scenario injection schedule of alternating short and long, once daily, dosing intervals (i.e., alternating intervals of 8 to 40 hours between doses). TRESIBA in this arm was dosed in the morning on Monday, Wednesday, and Friday and in the evening on Tuesday, Thursday, Saturday, and Sunday. Up to three of the following oral antidiabetes agents (metformin, sulfonylureas, glinides or thiazolidinediones) were administered as background therapy in both treatment arms.
The mean age of the trial population was 56 years and mean duration of diabetes was 11 years. 54% were male. 67% were White, 3% Black or African American. 11% were Hispanic. 6% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 29.6 kg/m².
At week 26, the difference in HbA1c reduction from baseline between TRESIBA at alternating times and insulin glargine U-100 was 0.04% with a 95% confidence interval of [-0.12%; 0.20%]. This comparison met the pre-specified non-inferiority margin (0.4%). See Table 12.
Table 12. Results at Week 26 in a Trial Comparing TRESIBA at Same and Alternating Times to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus on OAD (s)*:
| TRESIBA at the same time each day ± OAD (s)* | TRESIBA at alternating times ± OAD (s)* | Insulin glargine U-100 ± OAD (s)* | |
|---|---|---|---|
| N | 228 | 229 | 230 |
| HbA1c (%) | |||
| Baseline | 8.4 | 8.5 | 8.4 |
| End of trial | 7.3 | 7.2 | 7.1 |
| Adjusted mean change from baseline** | -1.03 | -1.17 | -1.21 |
| Estimated treatment difference [95%CI] TRESIBA alternating - Insulin glargine U-100 | 0.04 [-0.12;0.20] | ||
| Estimated treatment difference TRESIBA alternating – TRESIBA same | -0.13 | ||
| Proportion Achieving HbA1c <7% at Trial End | 40.8% | 38.9% | 43.9% |
| FPG (mg/dL) | |||
| Baseline | 158 | 162 | 163 |
| End of trial | 105 | 105 | 112 |
| Adjusted mean change from baseline | -54.2 | -55.0 | -47.5 |
| Daily insulin dose | |||
| Baseline mean | 21 U | 19 U | 19 U |
| Mean dose after 26 weeks | 45 U | 46 U | 44 U |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study G, there were 11.4% subjects for TRESIBA (both same time and alternating times) and 11.7% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
Study H: TRESIBA Administered at the Same Time Each Day in Combination with a Rapid-Acting Insulin Analog at Mealtimes in Adult Patients
The efficacy of TRESIBA was evaluated in a 52-week randomized, open-label, multicenter trial in 992 patients with type 2 diabetes mellitus inadequately controlled on premix insulin, bolus insulin alone, basal insulin alone, oral antidiabetic agents (OADs) alone or any combination thereof. Patients were randomized to TRESIBA once-daily with the main evening meal or insulin glargine U-100 once-daily according to the approved labeling. Insulin aspart was administered before each meal in both treatment arms. Up to two of the following oral antidiabetes agents (metformin or pioglitazone) were used as background therapy in both treatment arms.
The mean age of the trial population was 59 years and mean duration of diabetes was 14 years. 54% were male. 83% were White, 10% Black or African American. 12% were Hispanic. 12% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 32.2 kg/m².
At week 52, the difference in HbA1c reduction from baseline between TRESIBA and insulin glargine U-100 was 0.08% with a 95% confidence interval of [-0.05%; 0.21%] and met the pre-specified non-inferiority margin (0.4%). See Table 13.
Table 13. Results at Week 52 in a Trial Comparing TRESIBA to Insulin Glargine U-100 in Adult Patients with Type 2 Diabetes Mellitus Receiving Insulin Aspart at Mealtimes and OADs*:
| TRESIBA + Insulin aspart ± OAD (s)* | Insulin glargine U-100 + Insulin aspart ± OAD (s)* | |
|---|---|---|
| N | 744 | 248 |
| HbA1c (%) | ||
| Baseline | 8.3 | 8.4 |
| End of trial | 7.1 | 7.1 |
| Adjusted mean change from baseline** | -1.10 | -1.18 |
| Estimated treatment difference [95%CI] TRESIBA - Insulin glargine U-100 | 0.08 [-0.05;0.21] | |
| Proportion Achieving HbA1c <7% at Trial End | 49.5% | 50.0% |
| FPG (mg/dL) | ||
| Baseline | 166 | 166 |
| End of trial | 122 | 127 |
| Adjusted mean change from baseline | -40.6 | -35.3 |
| Daily basal insulin dose | ||
| Baseline mean | 42 U | 41 U |
| Mean dose after 52 weeks | 74 U | 67 U |
| Daily bolus insulin dose | ||
| Baseline mean | 33 U | 33 U |
| Mean dose after 52 weeks | 70 U | 73 U |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study H, there were 16.1% of subjects in the TRESIBA and 14.5% Insulin glargine arms for whom data was missing at the time of the HbA1c measurement.
Study I: TRESIBA Administered at Any Time Each Day as an Add-on to One or Two of the Following Oral Agents: Metformin, Sulfonylurea, or Pioglitazone in Adult Patients
The efficacy of TRESIBA was evaluated in a 26-week randomized, open-label, multicenter trial in 447 patients with type 2 diabetes mellitus inadequately controlled on one or more oral antidiabetic agent (OADs) at baseline. Patients were randomized to TRESIBA once-daily at any time of day or sitagliptin once-daily according to the approved labeling. One or two of the following oral antidiabetes agents (metformin, sulfonylurea or pioglitazone) were also administered in both treatment arms.
The mean age of the trial population was 56 years and mean duration of diabetes was 8 years. 59% were male. 61% were White, 8% Black or African American. 21% were Hispanic. 6% of patients had eGFR<60 mL/min/1.73m². The mean BMI was approximately 30.4 kg/m².
At the end of 26 weeks, TRESIBA provided greater reduction in mean HbA1c compared to sitagliptin (p<0.001). See Table 14.
Table 14. Results at Week 26 in a Trial Comparing TRESIBA to Sitagliptin in Adult Patients with Type 2 Diabetes Mellitus on OADs*:
| TRESIBA + OAD (s)* | Sitagliptin + OAD (s)* | |
|---|---|---|
| N | 225 | 222 |
| HbA1c (%) | ||
| Baseline | 8.8 | 9.0 |
| End of trial | 7.2 | 7.7 |
| Adjusted mean change from baseline** | -1.52 | -1.09 |
| Estimated treatment difference [95%CI] TRESIBA - Sitagliptin | -0.43 [-0.61;-0.24] 1 | |
| Proportion Achieving HbA1c <7% at Trial End | 40.9% | 27.9% |
| FPG (mg/dL) | ||
| Baseline | 170 | 179 |
| End of trial | 112 | 154 |
| Adjusted mean change from baseline | -61.4 | -22.3 |
| Daily insulin dose | ||
| Baseline mean | 10 U | N/A |
| Mean dose after 26 weeks | 43 U | N/A |
* OAD: oral antidiabetic agent
** The change from baseline to end of treatment visit in HbA1c was analyzed using ANOVA with treatment, region, sex, and anti-diabetic treatment at screening as fixed effects, and age and baseline HbA1c as covariates.
In Study I, there were 20.9% of subjects in the TRESIBA and 22.5% Sitagliptin arms for whom data was missing at the time of the HbA1c measurement.
1 p<0.001; 1-sided p-value evaluated at 2.5% level for superiority
14.4 Safety Outcomes Trial
DEVOTE (NCT01959529) Cardiovascular Outcomes Trial of TRESIBA Administered Once-Daily Between Dinner and Bedtime in Combination with Standard of Care in Subjects with Type 2 Diabetes and Atherosclerotic Cardiovascular Disease
DEVOTE was a multi-center, multi-national, randomized, double-blinded, active-controlled, treat-to-target, event-driven trial. 7,637 patients with inadequately controlled type 2 diabetes and atherosclerotic cardiovascular disease were randomized to either TRESIBA or insulin glargine U-100. Each was administered once-daily between dinner and bedtime in addition to standard of care for diabetes and cardiovascular disease for a median duration of 2 years.
Patients eligible to enter the trial were; 50 years of age or older and had established, stable, cardiovascular, cerebrovascular, peripheral artery disease, chronic kidney disease or NYHA class II and III heart failure (85% of the enrolled population) or were 60 years of age or older and had other specified risk factors for cardiovascular disease (15% of the enrolled population).
At baseline, demographic and disease characteristics were balanced between treatment groups. The mean age of the trial population was 65 years and the mean duration of diabetes was 16 years. The population was 63% male, 76% White 11% Black or African American, 10% Asian. 15% had Hispanic ethnicity. The mean HbA1c was 8.4% and the mean BMI was 33.6 kg/m². The baseline mean estimated glomerular filtration rate (eGFR) was 68 mL/min/1.73m². 41% of patients had eGFR 60-90 mL/min/1.73m²; 35% of patients had eGFR 30 to 60 mL/min/1.73 m² and 3% of patients had eGFR <30 mL/min/1.73 m². Previous history of severe hypoglycemia was not captured in the trial.
At baseline, patients treated their diabetes with oral antidiabetic drugs (72%) and with an insulin regimen (84%). Types of insulins included long acting insulin (60%), intermediate acting insulin (14%) short acting insulin (37%) and premixed insulin (10%). 16% of patients were insulin naive. The most common background oral antidiabetic drugs used at baseline were metformin (60%), sulfonylureas (29%) and DPP-4 inhibitors (12%).
During the trial, investigators could modify anti-diabetic and cardiovascular medications to achieve local standard of care treatment targets for lipids and blood pressure.
Cardiovascular Outcomes - Patients with T2DM and Atherosclerotic CVD:
The incidence of major cardiovascular events with TRESIBA was evaluated in DEVOTE. Subjects treated with TRESIBA had a similar incidence of major adverse cardiovascular events (MACE) when compared to those treated with insulin glargine U-100.
The primary endpoint in DEVOTE was time from randomization to the first occurrence of a 3-component major adverse cardiovascular event (MACE): cardiovascular death, non-fatal myocardial infarction, or non-fatal stroke. The trial was designed to exclude a pre-specified risk margin of 1.3 for the hazard ratio of MACE comparing TRESIBA to insulin glargine U-100. The primary outcome at end of trial was available for 98.2% of participants in each treatment group.
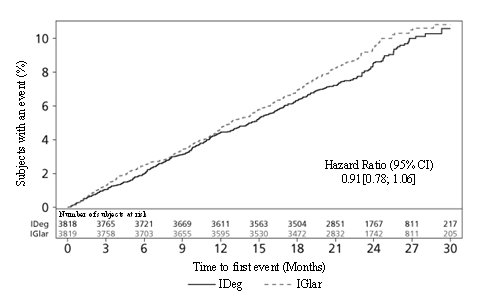
The time to first occurrence of MACE with TRESIBA as compared to insulin glargine U-100 was non-inferior (HR: 0.91; 95% CI [0.78;1.06]; see Figure 3). The results of the primary composite MACE endpoint and a summary of its individual components are shown in Table 15.
Table 15: Analysis of the Composite 3-point MACE and Individual Cardiovascular Endpoints in DEVOTE
| TRESIBA | Insulin glargine U-100 | ||||
|---|---|---|---|---|---|
| N | 3,818 | 3,819 | |||
| Number of Patients (%) | Rate per 100 PYO* | Number of Patients (%) | Rate per 100 PYO* | Hazard Ratio (95% CI) | |
| Composite of first event of CV death, non-fatal MI, or non-fatal stroke (3-Point MACE) | 325 (8.5) | 4.41 | 356 (9.3) | 4.86 | 0.91[0.78; 1.06] |
| CV death | 136 (3.6) | 1.85 | 142 (3.7) | 1.94 | |
| Non-fatal MI | 144 (3.8) | 1.95 | 169 (4.4) | 2.31 | |
| Non-fatal stroke | 71 (1.9) | 0.96 | 79 (2.1) | 1.08 | |
* PYO = patient-years of observation until first MACE, death, or trial discontinuation
Figure 3. Cumulative Event Probability for Time to First MACE in DEVOTE:
Hypoglycemia Outcomes - Patients with T2DM and Atherosclerotic CVD:
The pre-specified secondary endpoints of event and incidence rates of severe hypoglycemia were sequentially tested.
Severe hypoglycemia was defined as an episode requiring assistance of another person to actively administer carbohydrate, glucagon, or other resuscitative actions and during which plasma glucose concentration may not have been available, but where neurological recovery following the return of plasma glucose to normal was considered sufficient evidence that the event was induced by a low plasma glucose concentration.
The incidence of severe hypoglycemia was lower in the TRESIBA group as compared to the insulin glargine U-100 group (Table 16). Glycemic control between the two groups was similar at baseline and throughout the trial.
Table 16. Severe Hypoglycemic Episodes in Patients Treated with TRESIBA or Insulin Glargine U-100 in DEVOTE:
| TRESIBA | Insulin glargine U-100 | |
|---|---|---|
| N | 3,818 | 3,819 |
| Severe Hypoglycemia | ||
| Percent of patients with events | 4.9% | 6.6% |
| Estimated odds ratio [95%CI] TRESIBA/Insulin glargine U-100 | 0.73 [0.60; 0.89]* | |
| Events per 100 Patient Years of Observation | 3.70 | 6.25 |
| Estimated rate ratio [95%CI] TRESIBA/Insulin glargine U-100 | 0.60 [0.48; 0.76]* | |
* Test for superiority evaluated at 5% level for significance, (2-sided p<0.001)
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.