WAINUA Solution for injection Ref.[107295] Active ingredients: Eplontersen
Source: FDA, National Drug Code (US) Revision Year: 2023
12.1. Mechanism of Action
Eplontersen is an antisense oligonucleotide-GalNAc conjugate that causes degradation of mutant and wild-type TTR mRNA through binding to the TTR mRNA, which results in a reduction of serum TTR protein and TTR protein deposits in tissues.
12.2. Pharmacodynamics
In Study 1 [see Clinical Studies (14)], following administration of the recommended WAINUA dosage every 4 weeks to patients with hATTR amyloidosis, a decrease in serum TTR levels was observed at the first assessment and the (least square) mean serum TTR at Week 35 was reduced by 81% from baseline. Similar TTR reductions were observed across subgroups including Val30Met variant status, body weight, sex, age, or race.
Eplontersen also reduced the mean steady state serum vitamin A by 71% by Week 37 [see Warnings and Precautions (5.1)].
Cardiac Electrophysiology
At a dose 2.7-times the maximum recommended dose for WAINUA, clinically significant QTc interval prolongation was not observed.
12.3. Pharmacokinetics
The pharmacokinetic (PK) properties of WAINUA were evaluated following subcutaneous administration of single and multiple doses (once every 4 weeks) in healthy subjects and multiple doses (once every 4 weeks) in patients with hATTR amyloidosis.
Eplontersen Cmax and AUC showed a slightly greater than dose-proportional increase following single subcutaneous doses ranging from 45 to 120 mg (i.e., 1- to 2.7-times the recommended dose) in healthy volunteers.
Population estimates (mean ± SD) of steady state maximum concentrations (Cmax), and area under the curve (AUCτ) were 283 ± 152 ng/mL, and 2190 ± 689 ng/mL, respectively, following 45 mg monthly dosing in patients with hATTR amyloidosis. No accumulation of eplontersen Cmax and AUC was observed in repeated dosing (once every 4 weeks).
Absorption
Following subcutaneous administration, eplontersen is absorbed with the time to maximum plasma concentrations of approximately 2 hours, based on population estimates.
Distribution
Eplontersen is expected to distribute primarily to the liver and kidney cortex after subcutaneous dosing. Eplontersen is bound to human plasma proteins (>98%) in vitro. The population estimate for the apparent central volume of distribution is 12 L and the apparent peripheral volume of distribution is 11,100 L.
Elimination
The terminal elimination half-life is approximately 3 weeks.
Metabolism
Eplontersen is metabolized by endo- and exonucleases to short oligonucleotide fragments of varying sizes within the liver.
Excretion
The mean fraction of unchanged ASO eliminated in urine was less than 1% of the administered dose within 24 hours.
Specific Populations
Population pharmacokinetic and pharmacodynamic analysis showed no clinically meaningful differences in the pharmacokinetics or pharmacodynamics of eplontersen based on age, body weight, sex, race, Val30Met variant status, mild and moderate renal impairment (eGFR ≥30 to <90 mL/min), or mild hepatic impairment (total bilirubin ≤1 x ULN and AST >1 x ULN, or total bilirubin >1.0 to 1.5 x ULN and any AST). Eplontersen has not been studied in patients with severe renal impairment, end-stage renal disease, or in patients with moderate to severe hepatic impairment, or in patients with prior liver transplant.
Drug Interaction Studies
No clinical drug-drug interaction studies have been performed with eplontersen. In vitro studies show that eplontersen is not a substrate or inhibitor of transporters, does not interact with highly plasma protein bound drugs, and is not an inhibitor or inducer of cytochrome P450 (CYP) enzymes. Oligonucleotide therapeutics, including eplontersen, are not typically substrates of CYP enzymes. Therefore, eplontersen is not expected to cause or be affected by drug-drug interactions mediated through drug transporters, plasma protein binding or CYP enzymes.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis
Carcinogenicity studies have not been conducted with eplontersen.
Mutagenesis
Eplontersen was negative for genotoxicity in in vitro (bacterial reverse mutation, chromosomal aberration in Chinese hamster lung cells) and in vivo (mouse bone marrow micronucleus) assays.
Impairment of Fertility
Subcutaneous administration of eplontersen (0, 5, 25, or 75 mg/kg) or a mouse-specific surrogate (25 mg/kg) to male and female mice weekly prior to and during mating and continuing in females throughout the period of organogenesis resulted in no adverse effects on fertility.
14. Clinical Studies
The efficacy of WAINUA was demonstrated in a randomized, open-label, multicenter clinical trial in adult patients with polyneuropathy caused by hATTR amyloidosis (Study 1; NCT04136184). Patients were randomized in a 6:1 ratio to receive either 45 mg of WAINUA once every 4 weeks (N=144), or 284 mg of inotersen once per week (N=24), respectively, as subcutaneous injections. Ninety-seven percent of WAINUA-treated patients and 83% of inotersen-treated patients completed at least 35 weeks of the assigned treatment.
Efficacy assessments were based on a comparison of the WAINUA arm of Study 1 with an external placebo group (N=60) in another study (NCT01737398) composed of a comparable population of adult patients with polyneuropathy caused by hATTR amyloidosis.
The efficacy endpoints were the change from baseline to Week 35 in the modified Neuropathy Impairment Scale+7 (mNIS+7) composite score and the change from baseline to Week 35 in the Norfolk Quality of Life-Diabetic Neuropathy (QoL-DN) total score.
The mNIS+7 is an objective assessment of neuropathy and comprises the neuropathy impairment score (NIS) and Modified +7 composite scores. In the version of the mNIS+7 used in the trial, the NIS objectively measures deficits in cranial nerve function, muscle strength, reflexes, and sensations, and the Modified +7 assesses heart rate response to deep breathing, quantitative sensory testing (touch-pressure and heat-pain), and peripheral nerve electrophysiology. The validated version of the mNIS+7 score used in the trial has a range of -22.3 to 346.3 points, with higher scores representing a greater severity of disease.
The clinical meaningfulness of effects on the mNIS+7 was assessed by the change from baseline to Week 35 in Norfolk Quality of Life-Diabetic Neuropathy (QoL-DN) total score. The Norfolk QoL-DN scale is a patient-reported assessment that evaluates the subjective experience of neuropathy in the following domains: physical functioning/large fiber neuropathy, activities of daily living, symptoms, small fiber neuropathy, and autonomic neuropathy. The version of the Norfolk QoL-DN that was used in the trial has a range from -4 to 136 points, with higher scores representing greater impairment.
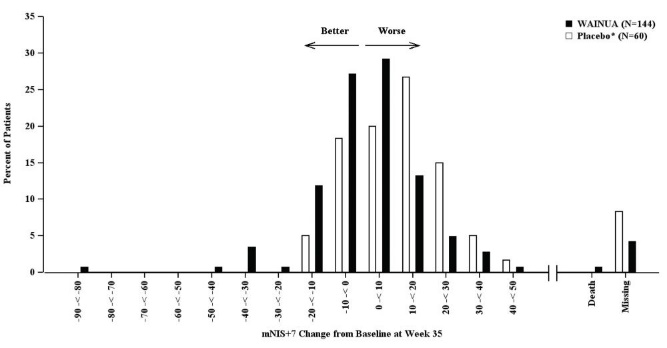
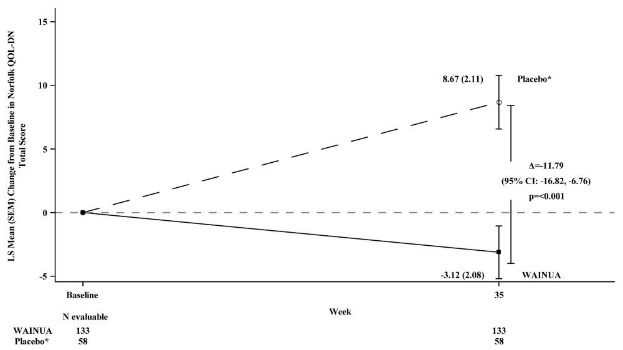
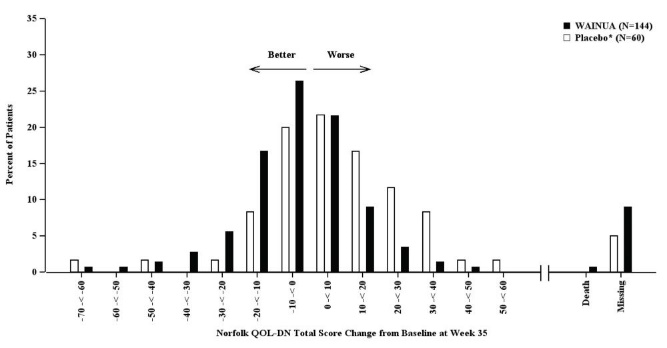
Treatment with WAINUA resulted in statistically significant improvements in the mNIS+7 and the Norfolk QoL-DN total scores, compared to the external placebo control (p<0.001) at Week 35 (Table 2, Figures 1 and 3). The distributions of changes in mNIS+7 and Norfolk QoL-DN scores from baseline to Week 35 by percent of patients in each category are shown in Figure 2 and Figure 4, respectively.
Table 2. Clinical Efficacy Results (Comparison of WAINUA Treatment in Study 1 to an External Placebo Control*):
| Endpoint | Baseline, Mean (SD) | Change from Baseline to Week 35, LS Mean (SEM) | Treatment Difference LS Mean (95% CI) | p-value | |||
|---|---|---|---|---|---|---|---|
| WAINUA N=140 (Study 1) | Placebo* N=59 (NCT01737398) | WAINUA (Study 1) | Placebo* (NCT01737398) | WAINUA- Placebo* | |||
| mNIS+7† | 79.6 (42.3) | 74.1 (39.0) | 0.2 (1.9) | 9.2 (1.9) | -9.0 (-13.5, -4.5) | <0.001 | |
| Norfolk QOL-DN† | 43.5 (26.3) | 48.6 (27.0) | -3.1 (2.1) | 8.7 (2.1) | -11.8 (-16.8, -6.8) | <0.001 | |
CI = confidence interval; LS mean = least squares mean; mNIS = modified Neuropathy Impairment Score; QoL-DN = Quality of Life-Diabetic Neuropathy; SD = standard deviation; SEM = standard error of the mean.
* External placebo group from another randomized controlled trial (NCT01737398).
† Based on an analysis of covariance (ANCOVA) model. Patients with a missing mNIS+7 or Norfolk QoL-DN at Week 35 had values multiply imputed using an imputation model.
Figure 1. Change from Baseline in mNIS+7 at Week 35 (Comparison of WAINUA Treatment in Study 1 to an External Placebo Control*):
* External placebo group from another randomized controlled trial (NCT01737398).
Based on an analysis of covariance (ANCOVA) model. Patients with a missing mNIS+7 at Week 35 had values multiply imputed using an imputation model.
Figure 2. Histogram of mNIS+7 Change from Baseline at Week 35 (Comparison of WAINUA Treatment in Study 1 to an External Placebo Control*):
* External placebo group from another randomized controlled trial (NCT01737398).
Categories are mutually exclusive; patients who died before Week 35 are summarized in the Death category only; missing mNIS+7 values not imputed; patients with missing values at Week 35 presented in the Missing category.
Figure 3. Change from Baseline in Norfolk QoL-DN Total Score at Week 35 (Comparison of WAINUA Treatment in Study 1 to an External Placebo Control*):
* External placebo group from another randomized controlled trial (NCT01737398).
Based on an analysis of covariance (ANCOVA) model. Patients with a missing Norfolk QoL-DN at Week 35 had values multiply imputed using an imputation model.
Figure 4. Histogram of Norfolk QoL-DN Total Score Change from Baseline at Week 35 (Comparison of WAINUA Treatment in Study 1 to an External Placebo Control*):
* External placebo group from another randomized controlled trial (NCT01737398).
Categories are mutually exclusive; patients who died before Week 35 are summarized in the Death category only; missing Norfolk QoL-DN values not imputed; patients with missing values at Week 35 presented in the Missing category.
Patients receiving WAINUA experienced similar improvements relative to those in the external placebo in mNIS+7, and Norfolk QoL-DN score across subgroups including age, sex, race, region, Val30Met variant status, and disease stage.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.