WYOST Solution for injection Ref.[109628] Active ingredients: Denosumab
Source: FDA, National Drug Code (US) Revision Year: 2024
12.1. Mechanism of Action
Denosumab products bind to RANKL, a transmembrane or soluble protein essential for the formation, function, and survival of osteoclasts, the cells responsible for bone resorption, thereby modulating calcium release from bone. Increased osteoclast activity, stimulated by RANKL, is a mediator of bone pathology in solid tumors with osseous metastases. Similarly, giant cell tumors of bone consist of stromal cells expressing RANKL and osteoclast-like giant cells expressing RANK receptor, and signaling through the RANK receptor contributes to osteolysis and tumor growth. Denosumab products prevent RANKL from activating its receptor, RANK, on the surface of osteoclasts, their precursors, and osteoclast-like giant cells.
12.2. Pharmacodynamics
In patients with breast cancer and bone metastases, the median reduction in uNTx/Cr was 82% within 1 week following initiation of denosumab 120 mg administered subcutaneously. In Studies 20050136, 20050244, and 20050103, the median reduction in uNTx/Cr from baseline to Month 3 was approximately 80% in 2075 denosumab-treated patients.
In a phase 3 study of patients with newly diagnosed multiple myeloma who received subcutaneous doses of denosumab 120 mg every 4 weeks (Q4W), median reductions in uNTx/Cr of approximately 75% were observed by week 5. Reductions in bone turnover markers were maintained, with median reductions of 74% to 79% for uNTx/Cr from weeks 9 to 49 of continued 120 mg Q4W dosing.
In adult and skeletally mature adolescent patients with giant cell tumor of bone who received subcutaneous doses of denosumab 120 mg Q4W with a 120 mg loading dose on Days 8 and 15, median reductions in uNTx/Cr from baseline were 84% at Week 13 and 82% at Week 25.
12.3. Pharmacokinetics
Following subcutaneous administration, bioavailability was 62%. Denosumab displayed nonlinear pharmacokinetics at doses below 60 mg, but approximately dose-proportional increases in exposure at higher doses.
With multiple subcutaneous doses of 120 mg once every 4 weeks, up to 2.8-fold accumulation in serum denosumab concentrations was observed and steady-state was achieved by 6 months. A mean (± standard deviation) serum steady-state trough concentration of 20.5 (± 13.5) mcg/mL was achieved by 6 months. The mean elimination half-life was 28 days.
In patients with newly diagnosed multiple myeloma who received 120 mg every 4 weeks, denosumab concentrations appear to reach steady-state by month 6. In patients with giant cell tumor of bone, after administration of subcutaneous doses of 120 mg once every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy, mean (± standard deviation) serum trough concentrations on Day 8, 15, and one month after the first dose were 19.0 (± 24.1), 31.6 (± 27.3), 36.4 (± 20.6) mcg/mL, respectively. Steadystate was achieved in 3 months after initiation of treatment with a mean serum trough concentration of 23.4 (± 12.1) mcg/mL.
Special Populations
Body Weight: A population pharmacokinetic analysis was performed to evaluate the effects of demographic characteristics. Denosumab clearance and volume of distribution were proportional to body weight. The steady-state exposure following repeat subcutaneous administration of 120 mg every 4 weeks to 45 kg and 120 kg subjects were, respectively, 48% higher and 46% lower than exposure of the typical 66 kg subject.
Age, Gender and Race: The pharmacokinetics of denosumab was not affected by age, gender, and race.
Pediatrics: In skeletally-mature adolescent patients (12 to 16 years of age) with giant cell tumor of bone (GCTB) who received 120 mg every 4 weeks with a 120 mg loading dose on Days 8 and 15, the pharmacokinetics of denosumab were comparable to those observed in adult patients with GCTB.
Hepatic Impairment: No clinical trials have been conducted to evaluate the effect of hepatic impairment on the pharmacokinetics of denosumab products.
Renal Impairment: In clinical trials of 87 patients with varying degrees of renal dysfunction, including patients on dialysis, the degree of renal impairment had no effect on the pharmacokinetics and pharmacodynamics of denosumab [see Use in Specific Populations (8.6)].
Drug Interactions
No formal drug-drug interaction trials have been conducted with denosumab. There was no evidence that various anticancer treatments affected denosumab systemic exposure and pharmacodynamic effect. Serum denosumab concentrations at 1 and 3 months and reductions in the bone turnover marker uNTx/Cr (urinary N-terminal telopeptide corrected for creatinine) at 3 months were similar in patients with and without prior intravenous bisphosphonate therapy and were not altered by concomitant chemotherapy and/or hormone therapy.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
The carcinogenic potential of denosumab products has not been evaluated in long-term animal studies. The genotoxic potential of denosumab products has not been evaluated.
Denosumab products have no effect on female fertility or male reproductive organs in monkeys at doses that were 6.5- to 25-fold higher than the recommended human dose of 120 mg subcutaneously administered once every 4 weeks, based on body weight (mg/kg).
13.2. Animal Toxicology and/or Pharmacology
Denosumab is an inhibitor of osteoclastic bone resorption via inhibition of RANKL.
Because the biological activity of denosumab in animals is specific to nonhuman primates, evaluation of genetically engineered (knockout) mice or use of other biological inhibitors of the RANK/RANKL pathway, OPG-Fc and RANK-Fc, provided additional information on the pharmacodynamic properties of denosumab. RANK/RANKL knockout mice exhibited absence of lymph node formation, as well as an absence of lactation due to inhibition of mammary gland maturation (lobulo-alveolar gland development during pregnancy). Neonatal RANK/RANKL knockout mice exhibited reduced bone growth and lack of tooth eruption. A corroborative study in 2-week-old rats given the RANKL inhibitor OPG-Fc also showed reduced bone growth, altered growth plates, and impaired tooth eruption. These changes were partially reversible in this model when dosing with the RANKL inhibitors was discontinued.
14. Clinical Studies
14.1 Bone Metastasis from Solid Tumors
The safety and efficacy of denosumab for the prevention of skeletal-related events in patients with bone metastases from solid tumors was demonstrated in three international, randomized (1:1), double-blind, activecontrolled, noninferiority trials comparing denosumab with zoledronic acid. In all three trials, patients were randomized to receive denosumab 120 mg subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In each trial, the main outcome measure was demonstration of noninferiority of time to first skeletal-related event (SRE) as compared to zoledronic acid. Supportive outcome measures were superiority of time to first SRE and superiority of time to first and subsequent SRE; testing for these outcome measures occurred if the main outcome measure was statistically significant. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20050136 (NCT00321464) enrolled 2046 patients with advanced breast cancer and bone metastasis. Randomization was stratified by a history of prior SRE (yes or no), receipt of chemotherapy within 6 weeks prior to randomization (yes or no), prior oral bisphosphonate use (yes or no), and region (Japan or other countries). Forty percent of patients had a previous SRE, 40% received chemotherapy within 6 weeks prior to randomization, 5% received prior oral bisphosphonates, and 7% were enrolled from Japan. Median age was 57 years, 80% of patients were White, and 99% of patients were women. The median number of doses administered was 18 for denosumab and 17 for zoledronic acid.
Study 20050244 (NCT00330759) enrolled 1776 adults with solid tumors other than breast and castrate-resistant prostate cancer with bone metastasis and multiple myeloma. Randomization was stratified by previous SRE (yes or no), systemic anticancer therapy at time of randomization (yes or no), and tumor type (non-small cell lung cancer, myeloma, or other). Eighty-seven percent were receiving systemic anticancer therapy at the time of randomization, 52% had a previous SRE, 64% of patients were men, 87% were White, and the median age was 60 years. A total of 40% of patients had non-small cell lung cancer, 10% had multiple myeloma, 9% had renal cell carcinoma, and 6% had small cell lung cancer. Other tumor types each comprised less than 5% of the enrolled population. The median number of doses administered was 7 for both denosumab and zoledronic acid.
Study 20050103 (NCT00321620) enrolled 1901 men with castrate-resistant prostate cancer and bone metastasis. Randomization was stratified by previous SRE, PSA level (less than 10 ng/mL or 10 ng/mL or greater) and receipt of chemotherapy within 6 weeks prior to randomization (yes or no). Twenty-six percent of patients had a previous SRE, 15% of patients had PSA less than 10 ng/mL, and 14% received chemotherapy within 6 weeks prior to randomization. Median age was 71 years and 86% of patients were White. The median number of doses administered was 13 for denosumab and 11 for zoledronic acid.
Denosumab delayed the time to first SRE following randomization as compared to zoledronic acid in patients with breast or castrate-resistant prostate cancer (CRPC) with osseous metastases (Table 2). In patients with bone metastasis due to other solid tumors or lytic lesions due to multiple myeloma, denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization.
Overall survival and progression-free survival were similar between arms in all three trials.
Table 2. Efficacy Results for Denosumab Compared to Zoledronic Acid:
| Study 20050136 Metastatic Breast Cancer | Study 20050244 Metastatic Solid Tumors or Multiple Myeloma | Study 20050103 Metastatic CRPCa | ||||
|---|---|---|---|---|---|---|
| Denosumab N = 1026 | Zoledronic Acid N = 1020 | Denosumab N = 886 | Zoledronic Acid N = 890 | Denosumab N = 950 | Zoledronic Acid N = 951 | |
| First On-study SRE | ||||||
| Number of Patients who had SREs (%) | 315 (30.7) | 372 (36.5) | 278 (31.4) | 323 (36.3) | 341 (35.9) | 386 (40.6) |
| Components of First SRE | ||||||
| Radiation to Bone | 82 (8.0) | 119 (11.7) | 119 (13.4) | 144 (16.2) | 177 (18.6) | 203 (21.3) |
| Pathological Fracture | 212 (20.7) | 238 (23.3) | 122 (13.8) | 139 (15.6) | 137 (14.4) | 143 (15.0) |
| Surgery to Bone | 12 (1.2) | 8 (0.8) | 13 (1.5) | 19 (2.1) | 1 (0.1) | 4 (0.4) |
| Spinal Cord Compression | 9 (0.9) | 7 (0.7) | 24 (2.7) | 21 (2.4) | 26 (2.7) | 36 (3.8) |
| Median Time to SRE (months) | NRb | 26.4 | 20.5 | 16.3 | 20.7 | 17.1 |
| Hazard Ratio (95% CI) | 0.82 (0.71, 0.95) | 0.84 (0.71, 0.98) | 0.82 (0.71, 0.95) | |||
| Noninferiority p-value | < 0.001 | < 0.001 | < 0.001 | |||
| Superiority p-valuec | 0.010 | 0.060 | 0.008 | |||
| First and Subsequent SREd | ||||||
| Mean Number/Patient | 0.46 | 0.60 | 0.44 | 0.49 | 0.52 | 0.61 |
| Rate Ratio (95% CI) | 0.77 (0.66, 0.89) | 0.90 (0.77, 1.04) | 0.82 (0.71, 0.94) | |||
| Superiority p-valuee | 0.001 | 0.145 | 0.009 | |||
a CRPC = castrate-resistant prostate cancer.
b NR = not reached. c Superiority testing performed only after denosumab demonstrated to be noninferior to zoledronic acid within trial.
d All skeletal events postrandomization; new events defined by occurrence ≥ 21 days after preceding event.
e Adjusted p-values are presented.
14.2 Multiple Myeloma
The efficacy of denosumab for the prevention of skeletal-related events in newly diagnosed multiple myeloma patients with treatment through disease progression, was evaluated in Study 20090482 (NCT01345019), an international, randomized (1:1), double-blind, active-controlled, noninferiority trial comparing denosumab with zoledronic acid. In this trial, patients were randomized to receive denosumab 120 mg subcutaneously every 4 weeks or 4 mg zoledronic acid intravenously (IV) every 4 weeks (dose adjusted for reduced renal function). Patients with creatinine clearance less than 30 mL/min were excluded. In this trial, the main efficacy outcome measure was noninferiority of time to first skeletal-related event (SRE). Additional efficacy outcome measures were superiority of time to first SRE, time to first and subsequent SRE, and overall survival. An SRE was defined as any of the following: pathologic fracture, radiation therapy to bone, surgery to bone, or spinal cord compression.
Study 20090482 enrolled 1718 newly diagnosed multiple myeloma patients with bone lesions. Randomization was stratified by a history of prior SRE (yes or no), the anti-myeloma agent being utilized/planned to be utilized in first-line therapy (novel therapy-based or non-novel therapy-based [novel therapies include bortezomib, lenalidomide, or thalidomide]), intent to undergo autologous PBSC transplantation (yes or no), stage at diagnosis (International Staging System I or II or III) and region Japan (yes or no). At study enrollment, 96% of the patients were receiving or planning to receive novel therapy-based first-line anti-myeloma therapy, 55% of the patients intended to undergo autologous PBSC transplantation, 61% of patients had a previous SRE, 32% were at ISS stage I, 38% were at ISS stage II and 29% were at ISS Stage III, and 2% were enrolled from Japan. Median age was 63 years, 82% of patients were White, and 46% of patients were women. The median number of doses administered was 16 for denosumab and 15 for zoledronic acid.
Denosumab was noninferior to zoledronic acid in delaying the time to first SRE following randomization (HR = 0.98, 95% CI, 0.85-1.14). The results for overall survival (OS) were comparable between denosumab and zoledronic acid treatment groups with a hazard ratio of 0.90 (95% CI: 0.70, 1.16).
Table 3. Efficacy Results for Denosumab Compared to Zoledronic Acid:
| Study 20090482 Multiple Myeloma | ||
|---|---|---|
| Denosumab N = 859 | Zoledronic Acid N = 859 | |
| First On-study SRE | ||
| Number of Patients who had SREs (%) | 376 (43.8) | 383 (44.6) |
| Components of First SRE | ||
| Radiation to Bone | 47 (5.5) | 62 (7.2) |
| Pathological Fracture | 342 (39.8) | 338 (39.3) |
| Surgery to Bone | 37 (4.3) | 48 (5.6) |
| Spinal Cord Compression | 6 (0.7) | 4 (0.5) |
| Median Time to SRE (months) (95% CI) | 22.8 (14.7, NEa) | 24 (16.6, 33.3) |
| Hazard Ratio (95% CI) | 0.98 (0.85, 1.14) | |
a NE = not estimable
14.3 Giant Cell Tumor of Bone
The safety and efficacy of denosumab for the treatment of giant cell tumor of bone in adults or skeletally mature adolescents were demonstrated in two open-label trials [Study 20040215 (NCT00396279) and Study 20062004 (NCT00680992)] that enrolled patients with histologically confirmed measurable giant cell tumor of bone that was either recurrent, unresectable, or for which planned surgery was likely to result in severe morbidity. Patients received denosumab 120 mg subcutaneously every 4 weeks with a loading dose on Days 8 and 15 of the first cycle of therapy. Patients who discontinued denosumab then entered the safety follow-up phase for a minimum of 60 months. Retreatment with denosumab while in safety follow-up was allowed for patients who initially demonstrated a response to denosumab (e.g., in the case of recurrent disease).
Study 20040215 was a single-arm, pharmacodynamic, and proof of concept trial conducted in 37 adult patients with unresectable or recurrent giant cell tumor of bone. Patients were required to have histologically confirmed giant cell tumor of bone and radiologic evidence of measurable disease from a computed tomography (CT) or magnetic resonance imaging (MRI) obtained within 28 days prior to study enrollment. Patients enrolled in Study 20040215 underwent CT or MRI assessment of giant cell tumor of bone at baseline and quarterly during denosumab treatment.
Study 20062004 was a parallel-cohort, proof of concept, and safety trial conducted in 535 adult or skeletally mature adolescent patients with histologically confirmed giant cell tumor of bone and evidence of measurable active disease. Study 20062004 enrolled 19 patients who were 12-16 years of age [see Use in Specific Populations (8.4)]. Patients enrolled into one of three cohorts: Cohort 1 enrolled 268 patients with surgically unsalvageable disease (e.g., sacral or spinal sites of disease, or pulmonary metastases); Cohort 2 enrolled 252 patients with surgically salvageable disease where the investigator determined that the planned surgery was likely to result in severe morbidity (e.g., joint resection, limb amputation, or hemipelvectomy); Cohort 3 enrolled 15 patients who previously participated in Study 20040215. Patients underwent imaging assessment of disease status at intervals determined by their treating physician.
A retrospective interim analysis concluded by an independent review committee evaluated objective response in 187 patients enrolled and treated in Study 20040215 and Study 20062004 for whom baseline and at least one post-baseline radiographic assessment were available (27 of 37 patients enrolled in Study 20040215 and 160 of 270 patients enrolled in Cohorts 1 and 2 of Study 20062004). The primary efficacy outcome measure was objective response rate using Response Evaluation Criteria in Solid Tumors (RECIST) v 1.1.
The overall objective response rate (RECIST 1.1) was 25% (95% CI: 19, 32). All responses were partial responses. The estimated median time to response was 3 months. In the 47 patients with an objective response, the median duration of follow-up was 20 months (range: 2-44 months), and 51% (24/47) had a duration of response lasting at least 8 months. Three patients experienced disease progression following an objective response.
14.4 Hypercalcemia of Malignancy
The safety and efficacy of denosumab was demonstrated in an open-label, single-arm trial [Study 20070315 (NCT00896454)] that enrolled 33 patients with hypercalcemia of malignancy (with or without bone metastases) refractory to treatment with intravenous bisphosphonate therapy. Patients received denosumab subcutaneously every 4 weeks with additional 120 mg doses on Days 8 and 15 of the first month of therapy.
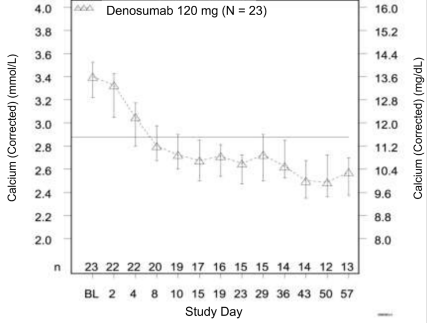
In this trial, refractory hypercalcemia of malignancy was defined as an albumin-corrected calcium of > 12.5 mg/dL (3.1 mmol/L) despite treatment with intravenous bisphosphonate therapy in 7-30 days prior to initiation of denosumab therapy. The primary outcome measure was the proportion of patients achieving a response, defined as corrected serum calcium (CSC) ≤ 11.5 mg/dL (2.9 mmol/L), within 10 days after denosumab administration. Efficacy data are summarized in Figure 1 and Table 4. Concurrent chemotherapy did not appear to affect response to denosumab.
Figure 1. Corrected Serum Calcium by Visit in Responders (Median and Interquartile Range):
N = Number of responders who received ≥ 1 dose of investigational product
n = Number of responders who had no missing data at baseline and the time point of interest
Table 4. Efficacy in Patients with Hypercalcemia of Malignancy Refractory to Bisphosphonate Therapy:
| N = 33 | Proportion () (95 CI) | |
|---|---|---|
| All Responders (CSC ≤ 11.5 mg/dL) by Day 10 | 21 | 63.6 (45.1, 79.6) |
| All Responders by Day 57 | 23 | 69.7 (51.3, 84.4) |
| Complete Responders (CSC ≤ 10.8 mg/dL) by Day 10 | 12 | 36.4 (20.4, 54.9) |
| All Complete Responders by Day 57 | 21 | 63.6 (45.1, 79.6) |
Median time to response (CSC ≤ 11.5 mg/dL) was 9 days (95% CI: 8, 19), and the median duration of response was 104 days (95% CI: 7, not estimable). Median time to complete response (CSC ≤ 10.8 mg/dL) was 23 days (95% CI: 9, 36), and the median duration of complete response was 34 days (95% CI: 1, 134).
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.