FASENRA Solution for injection Ref.[109450] Active ingredients: Benralizumab
Source: FDA, National Drug Code (US) Revision Year: 2024
12. Clinical Pharmacology
12.6 Immunogenicity
The observed incidence of anti-drug antibodies (ADA) is highly dependent on the sensitivity and specificity of the assay. Differences in assay methods preclude meaningful comparisons of the incidence of ADA in the studies described below with the incidence of ADA in other studies, including those of benralizumab and other products.
In adult and adolescent patients with asthma, treatment-emergent ADA response developed in 13% of patients treated with FASENRA at the recommended dosing regimen during the 48 to 56-week treatment period. A total of 12% of patients treated with FASENRA developed neutralizing antibodies. In patients 6 to 11 years with severe asthma and with an eosinophilic phenotype, the incidence of ADA to benralizumab during an open-label 48-week treatment period was comparable to adult and adolescent patients.
Anti-Drug Antibody Effects on Pharmacokinetics and Pharmacodynamics
Anti-benralizumab antibodies were associated with increased clearance of benralizumab and increased blood eosinophil levels in patients with high anti-drug antibody titers compared to antibody negative patients.
In the asthma trials in adults and adolescents, no evidence of an association of anti-drug antibodies with efficacy or safety was observed.
12.1. Mechanism of Action
Benralizumab is a humanized afucosylated, monoclonal antibody (IgG1, kappa) that directly binds to the alpha subunit of the human interleukin-5 receptor (IL-5Rα) with a dissociation constant of 11 pM. The IL-5 receptor is expressed on the surface of eosinophils and basophils. In an in vitro setting, the absence of fucose in the Fc domain of benralizumab facilitates binding (45.5 nM) to FcɣRIII receptors on immune effector cells, such as natural killer (NK) cells, leading to apoptosis of eosinophils and basophils through antibody-dependent cell-mediated cytotoxicity (ADCC).
Inflammation is an important component in the pathogenesis of asthma. Multiple cell types (e.g., mast cells, eosinophils, neutrophils, macrophages, lymphocytes) and mediators (e.g., histamine, eicosanoids, leukotrienes, cytokines) are involved in inflammation. Benralizumab, by binding to the IL-5Rα chain, reduces eosinophils through ADCC; however, the mechanism of benralizumab action in asthma has not been definitively established.
12.2. Pharmacodynamics
In the 52-week Phase 2 dose-ranging trial, asthma patients received 1 of 3 doses of benralizumab [2 mg (n=81), 20 mg (n=81), or 100 mg (n=222)] or placebo (n=222). All doses were administered every 4 weeks for the first 3 doses, followed by every 8 weeks thereafter. Median blood eosinophil levels at baseline were 310, 280, 190 and 190 cells/μL in the 2, 20, and 100 mg benralizumab and placebo groups, respectively. Dose-dependent reductions in blood eosinophils were observed. At the time of the last dose (Week 40), median blood eosinophil counts were 100, 50, 40, 170 cells/μL in the 2, 20, and 100 mg benralizumab and placebo groups, respectively.
A reduction in blood eosinophil counts was observed 24 hours post dosing in a Phase 2 trial.
In SIROCCO and CALIMA, following SC administration of benralizumab at the recommended dose blood eosinophils were reduced to a median absolute blood eosinophil count of 0 cells/μL [see Clinical Studies (14)]. This magnitude of reduction was observed at the first time point, 4 weeks of treatment, and was maintained throughout the treatment period.
Treatment with benralizumab was also associated with reductions in blood basophils, which was consistently observed across all clinical studies. In the Phase 2 dose-ranging trial, blood basophil counts were measured by flow cytometry. Median blood basophil counts were 45, 52, 46, and 40 cells/µL in the 2 mg, 20 mg and 100 mg benralizumab and placebo groups, respectively. At 52 weeks (12 weeks after the last dose), median blood basophil counts were 42, 18, 17, and 46 cells/µL in the 2 mg, 20 mg and 100 mg benralizumab and placebo groups, respectively.
In TATE, a 48-week trial with patients aged 6 to 11 years who had severe asthma, and with an eosinophilic phenotype [see Use in Specific Populations (8.4)], the magnitude of blood eosinophil reduction was similar to that observed in adults and adolescents. Median blood eosinophil levels at baseline were 400 and 340 cells/µL in patients weighing <35 kg and ≥35 kg, respectively. Across all post-dose time points, median eosinophil counts reduced to 10 to 20 cells/µL in patients weighing <35 kg, and to 20 to 30 cells/µL in patients weighing ≥35 kg. Blood eosinophil reduction was observed at the first time point, 4 weeks of treatment, and was maintained throughout the treatment period.
12.3. Pharmacokinetics
The pharmacokinetics of benralizumab was approximately dose-proportional in adult and adolescent patients with asthma following subcutaneous administration over a dose range of 20 to 200 mg.
Absorption
Following subcutaneous administration to patients with asthma, the absorption half-life was approximately 3.5 days. Based on population pharmacokinetic analysis, the estimated absolute bioavailability was approximately 59% and there was no clinically relevant difference in relative bioavailability in the administration to the abdomen, thigh, or arm.
Distribution
Based on population pharmacokinetic analysis, central and peripheral volume of distribution of benralizumab was 3.1 L and 2.5 L, respectively, for a 70 kg individual.
Elimination
From population pharmacokinetic analysis, benralizumab exhibited linear pharmacokinetics and no evidence of target receptor-mediated clearance pathway. The estimated typical systemic clearance (CL) for benralizumab was 0.29 L/d for a subject weighing 70 kg. Following subcutaneous administration, the elimination half-life was approximately 15.5 days.
Metabolism
Benralizumab is a humanized IgG1 monoclonal antibody that is degraded by proteolytic enzymes widely distributed in the body and not restricted to hepatic tissue.
Specific populations
Age
Based on population pharmacokinetic analysis, age did not affect benralizumab clearance.
Gender, Race
A population pharmacokinetics analysis indicated that there was no significant effect of gender and race on benralizumab clearance.
Patients with Renal impairment
No formal clinical studies have been conducted to investigate the effect of renal impairment on benralizumab. Based on population pharmacokinetic analysis, benralizumab clearance was comparable in subjects with creatinine clearance values between 30 and 80 mL/min and patients with normal renal function. There are limited data available in subjects with creatinine clearance values less than 30 mL/min; however, benralizumab is not cleared renally.
Patients with Hepatic impairment
No formal clinical studies have been conducted to investigate the effect of hepatic impairment on benralizumab. IgG monoclonal antibodies are not primarily cleared via hepatic pathway; change in hepatic function is not expected to influence benralizumab clearance. Based on population pharmacokinetic analysis, baseline hepatic function biomarkers (ALT, AST, and bilirubin) had no clinically relevant effect on benralizumab clearance.
Pediatric Patients
Benralizumab pharmacokinetics following subcutaneous administration of 10 mg or 30 mg in patients aged 6 to 11 years with severe asthma and with an eosinophilic phenotype asthma was investigated in the initial 16-week treatment phase of TATE, a 48-week open-label trial. Among patients aged 6 to 11 years weighing <35 kg who received 10 mg, the median trough concentration at Week 16 was similar to that of adults and adolescents who received 30 mg. Among patients aged 6 to 11 years weighing ≥35 kg who received 30 mg, the median trough concentration at Week 16 was 62% higher relative to adults and adolescents receiving the same dose, due to lower body weight in pediatric patients.
Drug Interaction Studies
No formal drug-drug interaction studies have been conducted.
Cytochrome P450 enzymes, efflux pumps and protein-binding mechanisms are not involved in the clearance of benralizumab. There is no evidence of IL-5Rα expression on hepatocytes and eosinophil depletion does not produce chronic systemic alterations of proinflammatory cytokines.
An effect of benralizumab on the pharmacokinetics of co-administered medications is not expected. Based on the population analysis, commonly co-administered medications had no effect on benralizumab clearance in patients with asthma.
13.1. Carcinogenesis, Mutagenesis, Impairment of Fertility
Long-term animal studies have not been performed to evaluate the carcinogenic potential of benralizumab. Published literature using animal models suggests that IL-5 and eosinophils are part of an early inflammatory reaction at the site of tumorigenesis and can promote tumor rejection. However, other reports indicate that eosinophil infiltration into tumors can promote tumor growth. Therefore, the malignancy risk in humans from an antibody that binds to IL-5Rα such as benralizumab is unknown.
Male and female fertility were unaffected based upon no adverse histopathological findings in the reproductive organs from cynomolgus monkeys treated with benralizumab for 9 months at IV doses up to 25 mg/kg or at SC doses of up to 30 mg/kg once every 2 weeks (approximately 400 and 270 times the MRHD on an AUC basis).
14. Clinical Studies
The efficacy of FASENRA was evaluated in two randomized, double-blind, parallel-group, placebo-controlled, exacerbation trials, SIROCCO (NCT01928771) and CALIMA (NCT01914757), for 48 and 56 weeks in duration, respectively. Furthermore, the effects of FASENRA in the reduction of oral corticosteroid use and effect on lung function were evaluated in clinical trials, ZONDA (NCT02075255) and a 12-week lung function trial (NCT02322775), respectively.
SIROCCO and CALIMA were randomized, double-blind, parallel-group, placebo-controlled, exacerbation trials in patients 12 years of age and older and 48 and 56 weeks in duration, respectively. The trials randomized a total of 2,510 patients. Patients were required to have a history of 2 or more asthma exacerbations requiring oral or systemic corticosteroid treatment in the past 12 months, ACQ-6 score of 1.5 or more at screening, and reduced lung function at baseline [pre-bronchodilator FEV1 below 80% in adults, and below 90% in adolescents] despite regular treatment with high dose inhaled corticosteroid (ICS) (SIROCCO) or with medium or high dose ICS (CALIMA) plus a long-acting beta agonist (LABA) with or without oral corticosteroids (OCS) and additional asthma controller medications. Patients were stratified by geography, age, and blood eosinophils count (≥300 cells/μL or <300 cells/μL). FASENRA administered once every 4 weeks for the first 3 doses, and then every 4 or 8 weeks thereafter as add-on to background treatment was evaluated compared to placebo.
All subjects continued their background asthma therapy throughout the duration of the trials.
ZONDA was a randomized, double-blind, parallel-group, OCS reduction trial in 220 adult patients with asthma. Patients were required treatment with daily OCS (7.5 to 40 mg per day) in addition to regular use of high-dose ICS and LABA with or without additional controller(s). The trial included an 8-week run-in period during which the OCS was titrated to the minimum effective dose without losing asthma control. For the purposes of the OCS dose titration, asthma control was assessed by the investigator based on a patient's FEV1, peak expiratory flow, nighttime awakenings, short-acting bronchodilator rescue medication use or any other symptoms that would require an increase in OCS dose. Baseline median OCS dose was similar across all treatment groups. Patients were required to have blood eosinophil counts greater than or equal to 150 cells/μL and a history of at least one exacerbation in the past 12 months. The baseline median OCS dose was 10 mg (range: 8 to 40 mg) for all 3 treatment groups (placebo, FASENRA every 4 weeks, and FASENRA every 4 weeks for the first 3 doses, and then once every 8 weeks).
While 2 dosing regimens were studied in SIROCCO, CALIMA, and ZONDA, the recommended dosing regimen is 30 mg FASENRA administered every 4 weeks for the first 3 doses, then every 8 weeks thereafter [see Dosage and Administration (2.1)].
Table 3. Demographics and Baseline Characteristics of Asthma Trials:
| Total Population | |||
|---|---|---|---|
| SIROCCO (N=1204) | CALIMA (N=1306) | ZONDA (N=220) | |
| Mean age (yr) | 49 | 49 | 51 |
| Female (%) | 66 | 62 | 61 |
| White (%) | 73 | 84 | 93 |
| Duration of asthma, median (yr) | 15 | 16 | 12 |
| Never smoked (%) | 80 | 78 | 79 |
| Mean baseline FEV1 pre-bronchodilator (L) | 1.67 | 1.76 | 1.85 |
| Mean baseline % predicted FEV1 | 57 | 58 | 60 |
| Mean post-SABA FEV1/FVC (%) | 66 | 65 | 62 |
| Mean baseline eosinophil count (cells/μL) | 472 | 472 | 575 |
| Mean number of exacerbations in previous year | 3 | 3 | 3 |
Exacerbations
The primary endpoint for SIROCCO and CALIMA was the rate of asthma exacerbations in patients with baseline blood eosinophil counts of greater than or equal to 300 cells/μL who were taking high-dose ICS and LABA. Asthma exacerbation was defined as a worsening of asthma requiring use of oral/systemic corticosteroids for at least 3 days, and/or emergency department visits requiring use of oral/systemic corticosteroids and/or hospitalization. For patients on maintenance oral corticosteroids, an asthma exacerbation requiring oral corticosteroids was defined as a temporary increase in stable oral/systemic corticosteroids for at least 3 days or a single depo-injectable dose of corticosteroids. In SIROCCO , 35% of patients receiving FASENRA experienced an asthma exacerbation compared to 51% on placebo. In CALIMA, 40% of patients receiving FASENRA experienced an asthma exacerbation compared to 51% on placebo (Table 4).
Table 4. Rate of Exacerbations, SIROCCO and CALIMA (ITT Population)*:
| Trial | Treatment | Exacerbations per year | ||
|---|---|---|---|---|
| Rate | Difference | Rate Ratio (95% CI) | ||
| All exacerbations | ||||
| SIROCCO | FASENRA† (n=267) | 0.74 | -0.78 | 0.49 (0.37, 0.64) |
| Placebo (n=267) | 1.52 | -- | -- | |
| CALIMA | FASENRA† (n=239) | 0.73 | -0.29 | 0.72 (0.54, 0.95) |
| Placebo (n=248) | 1.01 | -- | -- | |
| Exacerbations requiring hospitalization/emergency room visit | ||||
| SIROCCO | FASENRA† (n=267) | 0.09 | -0.16 | 0.37 (0.20, 0.67) |
| Placebo (n=267) | 0.25 | -- | -- | |
| CALIMA | FASENRA† (n=239) | 0.12 | 0.02 | 1.23 (0.64, 2.35) |
| Placebo (n=248) | 0.10 | -- | -- | |
| Exacerbations requiring hospitalization | ||||
| SIROCCO | FASENRA† (n=267) | 0.07 | -0.07 | 0.48 (0.22, 1.03) |
| Placebo (n=267) | 0.14 | -- | -- | |
| CALIMA | FASENRA† (n=239) | 0.07 | 0.02 | 1.48 (0.65, 3.37) |
| Placebo (n=248) | 0.05 | -- | -- | |
* Baseline blood eosinophil counts of greater than or equal to 300 cells/μL and taking high-dose ICS
† FASENRA 30 mg administered every 4 weeks for the first 3 doses, and every 8 weeks thereafter
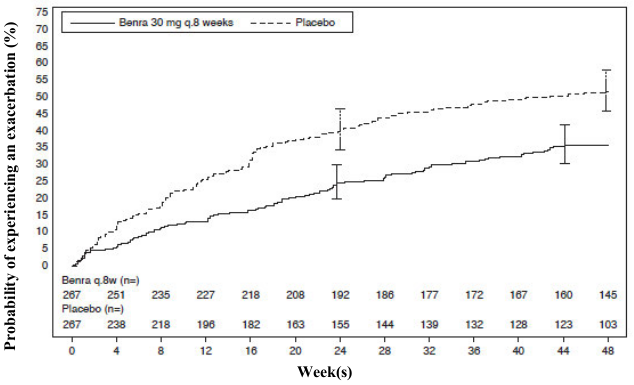
The time to first exacerbation was longer for the patients receiving FASENRA compared with placebo in SIROCCO (Figure 2). Similar findings were seen in CALIMA.
Figure 2. Kaplan-Meier Cumulative Incidence Curves for Time to First Exacerbation, SIROCCO:
Subgroup analyses from SIROCCO and CALIMA identified patients with a higher prior exacerbation history and baseline blood eosinophil count as potential predictors of improved treatment response. Reductions in exacerbation rates were observed irrespective of baseline peripheral eosinophil counts; however, patients with a baseline blood eosinophil count ≥300 cells/μL showed a numerically greater response than those with counts <300 cells/μL. In both trials patients with a history of 3 or more exacerbations within the 12 months prior to FASENRA randomization showed a numerically greater exacerbation response than those with fewer prior exacerbations.
Oral Corticosteroid Reduction
ZONDA evaluated the effect of FASENRA on reducing the use of maintenance oral corticosteroids in adult patients with asthma. The primary endpoint was percent reduction from baseline of the final OCS dose during Weeks 24 to 28, while maintaining asthma control (see definition of asthma control in trial description). Compared to placebo, patients receiving FASENRA achieved greater reductions in daily maintenance oral corticosteroid dose, while maintaining asthma control. The median percent reduction in daily OCS dose from baseline was 75% in patients receiving FASENRA (95% CI: 60, 88) compared to 25% in patients receiving placebo (95% CI: 0, 33). Reductions of 50% or higher in the OCS dose were observed in 48 (66%) patients receiving FASENRA compared to those receiving placebo 28 (37%). The proportion of patients with a mean final dose less than or equal to 5 mg at Weeks 24 to 28 was 59% for FASENRA and 33% for placebo (odds ratio 2.74, 95% CI: 1.41, 5.31). Only patients with an optimized baseline OCS dose of 12.5 mg or less were eligible to achieve a 100% reduction in OCS dose during the study. Of those patients, 52% (22 of 42) receiving FASENRA and 19% (8 of 42) on placebo achieved a 100% reduction in OCS dose. Exacerbations resulting in hospitalization and/or ER visit were also assessed as a secondary endpoint. In this 28-week trial, patients receiving FASENRA had 1 event while those on placebo had 14 events (annualized rate 0.02 and 0.32, respectively; rate ratio of 0.07, 95% CI: 0.01, 0.63).
Lung Function
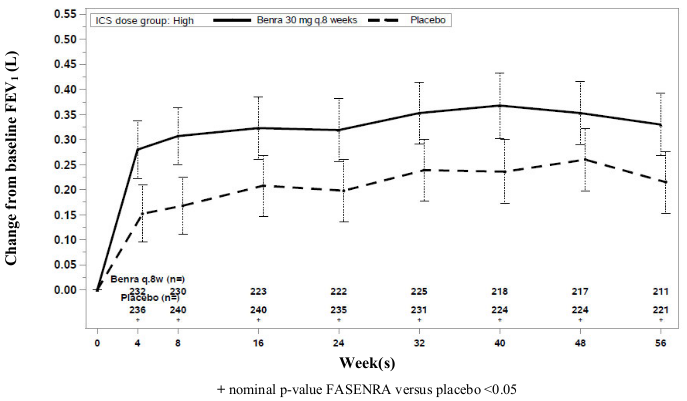
Change from baseline in mean FEV1 was assessed in SIROCCO, CALIMA, and ZONDA as a secondary endpoint. Compared with placebo, FASENRA provided consistent improvements over time in the mean change from baseline in FEV1 (Figure 3 and Table 5).
Figure 3. Mean Change from Baseline in Pre-Bronchodilator FEV1 (L), CALIMA:
Table 5. Change from Baseline in Mean Pre-Bronchodilator FEV1 (L) at End of Trial*:
| Trial | Difference from Placebo in Mean Change from Pre-Bronchodilator Baseline FEV1 (L) (95% CI) |
|---|---|
| SIROCCO | 0.159 (0.068, 0.249) |
| CALIMA | 0.116 (0.028, 0.204) |
| ZONDA | 0.112 (-0.033, 0.258) |
* Week 48 in SIROCCO, Week 56 in CALIMA, Week 28 in ZONDA.
Subgroup analyses also showed greater improvements in FEV1 in patients with higher baseline blood eosinophil counts and more frequent prior exacerbation history.
The clinical program for FASENRA also included a 12-week, randomized, double-blind, placebo-controlled lung function trial conducted in 211 adult patients with mild to moderate asthma. Patients were treated with placebo or benralizumab 30 mg SC every 4 weeks for 3 doses. Lung function, as measured by the change from baseline in FEV1 at Week 12 was improved in the benralizumab treatment group compared to placebo.
Patient Reported Outcomes
The Asthma Control Questionnaire-6 (ACQ-6) and Standardized Asthma Quality of Life Questionnaire for 12 Years and Older (AQLQ(S)+12) were assessed in SIROCCO, CALIMA, and ZONDA. The responder rate for both measures was defined as improvement in score of 0.5 or more as threshold at the end of SIROCCO, CALIMA, and ZONDA (48, 56, and 28 weeks, respectively). In SIROCCO, the ACQ-6 responder rate for FASENRA was 60% vs 50% placebo (odds ratio 1.55; 95% CI: 1.09, 2.19). In CALIMA, the ACQ-6 responder rate for the FASENRA was 63% vs 59% placebo (odds ratio 1.16; 95% CI: 0.80, 1.68). In SIROCCO, the responder rate for AQLQ(S)+12 for FASENRA was 57% vs 49% placebo (odds ratio 1.42; 95% CI: 0.99, 2.02), and in CALIMA, 60% FASENRA vs 59% placebo (odds ratio of 1.03; 95% CI: 0.70,1.51). Similar results were seen in ZONDA.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.