FRUZAQLA Hard capsule Ref.[107236] Active ingredients: Fruquintinib
Source: FDA, National Drug Code (US) Revision Year: 2023
Product description
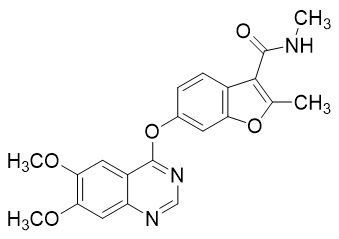
Fruquintinib is a kinase inhibitor with the chemical name 6-[(6,7-dimethoxyquinazolin-4-yl)oxy]-N,2-dimethyl-1-benzofuran-3-carboxamide. Its molecular formula is C21H19N3O5, which corresponds to a molecular weight of 393.39 g/mol.
Fruquintinib has the following chemical structure:
Fruquintinib is a white to off-white powder with a dissociation constant (pKa) of 2.78. The aqueous solubility of fruquintinib is pH-dependent with a solubility of 0.9 μg/mL at pH 6.8 that increases under acidic conditions to 129.9 μg/mL at pH 1.
FRUZAQLA (fruquintinib) capsules for oral administration contain 1 mg or 5 mg of fruquintinib. The inactive ingredients are corn starch, microcrystalline cellulose, and talc. The 1 mg capsule shell contains FD&C Yellow No. 5 (tartrazine), FD&C Yellow No. 6 (sunset yellow FCF), gelatin, and titanium dioxide. The 5 mg capsule shell contains FD&C Blue No. 1 (brilliant blue FCF), FD&C Red No. 40 (allura red AC), gelatin, and titanium dioxide. The printing ink for 1 mg and 5 mg capsules contains butanol, dehydrated alcohol, ferrosoferric oxide, isopropyl alcohol, potassium hydroxide, propylene glycol, purified water, shellac and strong ammonia solution.
| Dosage Forms and Strengths |
|---|
|
Capsules:
|
| How Supplied | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
Distributed by: Takeda Pharmaceuticals America, Inc., Lexington, MA 02421 |
Drugs
| Drug | Countries | |
|---|---|---|
| FRUZAQLA | Austria, Estonia, Spain, France, Italy, Lithuania, Romania, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.