STAXYN Tablet, orally disintegrating Ref.[11069] Active ingredients: Vardenafil
Source: FDA, National Drug Code (US) Revision Year: 2012
Product description
STAXYN is an oral therapy for the treatment of erectile dysfunction. This monohydrochloride salt of vardenafil is a selective inhibitor of cyclic guanosine monophosphate (cGMP)-specific PDE5.
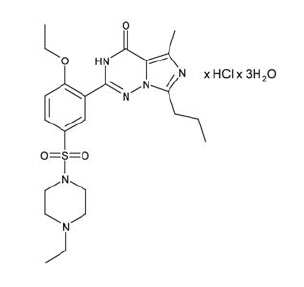
Vardenafil HCl is designated chemically as piperazine, 1-[[3-(1,4-dihydro-5-methyl-4-oxo-7-propylimidazo[5,1-f][1,2,4]triazin-2-yl)-4-ethoxyphenyl]sulfonyl]-4-ethyl-, monohydrochloride and has the following structural formula:
Vardenafil HCl is a nearly colorless, solid substance with a molecular weight of 579.1 g/mol and a solubility of 0.11 mg/mL in water.
STAXYN is formulated as white round orally disintegrating tablets with no debossing. Each tablet contains 11.85 mg vardenafil hydrochloride, which corresponds to 10 mg vardenafil, and the following inactive ingredients: aspartame, peppermint flavor, magnesium stearate, and Pharmaburst B2 (crospovidone, mannitol, silica colloidal hydrated, and sorbitol).
| Dosage Forms and Strengths |
|---|
|
STAXYN is available in 10 mg white, round, orally disintegrating tablets (not scored), no debossing. |
| How Supplied | ||||||
|---|---|---|---|---|---|---|
|
STAXYN (vardenafil HCl) are white, round orally disintegrating tablets with no debossing. STAXYN orally disintegrating tablets are packaged into foil blisterpacks and supplied as a 4 tablet unit or as a 40 tablet bulk pack.
In addition to the active ingredient, vardenafil, each tablet contains aspartame, peppermint flavor, magnesium stearate, and Pharmaburst B2 (crospovidone, mannitol, silica colloidal hydrated, and sorbitol). |
Drugs
| Drug | Countries | |
|---|---|---|
| STAXYN | Canada, United States |
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.