TRIPLIXAM Film-coated tablet Ref.[107764] Active ingredients: Amlodipine Indapamide Perindopril Perindopril, Amlodipine and Indapamide
Source: Health Sciences Authority (SG) Revision Year: 2023 Publisher: Les Laboratoires Servier – France <u>Manufacturer:</u> Servier (Ireland) Industries Ltd., Moneylands, Gorey Road, Arklow, Co. Wicklow – Ireland
4.3. Contraindications
- Dialysis patients.
- Patients with untreated decompensated heart failure.
- Severe renal impairment (creatinine clearance below 30 mL/min).
- Moderate renal impairment (creatinine clearance below 60 mL/min) for Triplixam doses containing 10mg/2.5mg of perindopril/indapamide combination (i.e., Triplixam 10mg/2.5mg/5mg and 10mg/2.5mg/10mg).
- Hypersensitivity to the active substances, to other sulfonamides, to dihydropyridine derivatives, any other ACE-inhibitor or to any of the excipients listed in section 6.1.
- History of angioedema (Quincke’s oedema) associated with previous ACE inhibitor therapy (see section 4.4).
- Hereditary/idiopathic angioedema.
- Second and third trimesters of pregnancy (see sections 4.4 and 4.6).
- Hepatic encephalopathy.
- Severe hepatic impairment.
- Hypokalaemia.
- Severe hypotension.
- Shock, including cardiogenic shock.
- Obstruction of the outflow-tract of the left ventricle (e.g. high grade aortic stenosis).
- Haemodynamically unstable heart failure after acute myocardial infarction.
- Concomitant use of Triplixam with aliskiren-containing products in patients with diabetes mellitus or renal impairment (GFR <60mL/min/1.73m²) (see sections 4.5 and 5.1).
- Concomitant use with sacubitril/valsartan therapy, Triplixam must not be initiated earlier than 36 hours after the last dose of sacubitril/valsartan (see sections 4.4 and 4.5).
- Extracorporeal treatments leading to contact of blood with negatively charged surfaces (see section 4.5).
- Significant bilateral renal artery stenosis or stenosis of the artery to a single functioning kidney (see section 4.4).
4.4. Special warnings and precautions for use
All warnings related to each component, as listed below, should apply also to the fixed combination of Triplixam.
Special warnings
Lithium
The combination of lithium and the combination of perindopril/indapamide is usually not recommended (see section 4.5).
Dual blockade of the renin-angiotensin-aldosterone system (RAAS)
There is evidence that the concomitant use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren increases the risk of hypotension, hyperkalaemia and decreased renal function (including acute renal failure). Dual blockade of RAAS through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is therefore not recommended (see sections 4.5 and 5.1).
If dual blockade therapy is considered absolutely necessary, this should only occur under specialist supervision and subject to frequent close monitoring of renal function, electrolytes and blood pressure.
ACE-inhibitors and angiotensin II receptor blockers should not be used concomitantly in patients with diabetic nephropathy.
Potassium-sparing drugs, potassium supplements or potassium-containing salt substitutes
The combination of perindopril and potassium-sparing drugs, potassium supplements or potassiumcontaining salt substitutes is usually not recommended (see section 4.5).
Neutropenia/agranulocytosis/thrombocytopenia/anaemia
Neutropenia/agranulocytosis, thrombocytopenia and anaemia have been reported in patients receiving ACE inhibitors. In patients with normal renal function and no other complicating factors, neutropenia occurs rarely. Perindopril should be used with extreme caution in patients with collagen vascular disease, immunosuppressant therapy, treatment with allopurinol or procainamide, or a combination of these complicating factors, especially if there is pre-existing impaired renal function. Some of these patients developed serious infections which in a few instances did not respond to intensive antibiotic therapy. If perindopril is used in such patients, periodical monitoring of white blood cell counts is advised and patients should be instructed to report any sign of infection (e.g. sore throat, fever) (see section 4.8).
Renovascular hypertension
There is an increased risk of hypotension and renal insufficiency when patient with bilateral renal artery stenosis or stenosis of the artery to a single functioning kidney are treated with ACE inhibitors (see section 4.3). Treatment with diuretics may be a contributory factor. Loss of renal function may occur with only minor changes in serum creatinine even in patients with unilateral renal artery stenosis.
Hypersensitivity/angioedema
Angioedema of the face, extremities, lips, tongue, glottis and/or larynx has been reported rarely in patients treated with angiotensin converting enzyme inhibitors, including perindopril. This may occur at any time during treatment. In such cases perindopril should be discontinued promptly and appropriate monitoring should be instituted to ensure complete resolution of symptoms prior to dismissing the patient. In those instances where swelling has been confined to the face and lips the condition generally resolved without treatment, although antihistamines have been useful in relieving symptoms.
Angioedema associated with laryngeal oedema may be fatal. Where there is involvement of the tongue, glottis or larynx, likely to cause airway obstruction, appropriate therapy, which may include subcutaneous epinephrine solution 1:1000 (0.3 mL to 0.5 mL) and/or measures to ensure a patent airway, should be administered promptly.
Black patients receiving ACE inhibitors have been reported to have a higher incidence of angioedema compared to non-blacks.
Patients with a history of angioedema unrelated to ACE inhibitor therapy may be at increased risk of angioedema while receiving an ACE inhibitor (see section 4.3).
Intestinal angioedema has been reported rarely in patients treated with ACE inhibitors. These patients presented with abdominal pain (with or without nausea or vomiting); in some cases there was no prior facial angioedema and C-1 esterase levels were normal. The angioedema was diagnosed by procedures including abdominal CT scan, or ultrasound or at surgery and symptoms resolved after stopping the ACE inhibitor. Intestinal angioedema should be included in the differential diagnosis of patients on ACE inhibitors presenting with abdominal pain.
The combination of perindopril with sacubitril/valsartan is contraindicated due to the increased risk of angioedema (see section 4.3). Sacubitril/valsartan must not be initiated until 36 hours after taking the last dose of perindopril therapy. If treatment with sacubitril/valsartan is stopped, perindopril therapy must not be initiated until 36 hours after the last dose of sacubitril/valsartan (see sections 4.3 and 4.5).
Concomitant use of ACE inhibitors with NEP inhibitors (e.g. racecadotril), mTOR inhibitors (e.g. sirolimus, everolimus, temsirolimus) and gliptins (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin) may lead to an increased risk of angioedema (e.g. swelling of the airways or tongue, with or without respiratory impairment) (see section 4.5). Caution should be used when starting racecadotril, mTOR inhibitors (e.g. sirolimus, everolimus, temsirolimus) and gliptins (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin) in a patient already taking an ACE inhibitor.
Anaphylactoid reactions during desensitization
There have been isolated reports of patients experiencing sustained, life-threatening anaphylactoid reactions while receiving ACE inhibitors during desensitisation treatment with hymenoptera (bees, wasps) venom. ACE inhibitors should be used with caution in allergic patients treated with desensitisation, and avoided in those undergoing venom immunotherapy. However these reactions could be prevented by temporary withdrawal of ACE inhibitor for at least 24 hours before treatment in patients who require both ACE inhibitors and desensitisation.
Anaphylactoid reactions during LDL apheresis
Rarely, patients receiving ACE inhibitors during low density lipoprotein (LDL)-apheresis with dextran sulphate have experienced life-threatening anaphylactoid reactions. These reactions were avoided by temporarily withholding ACE-inhibitor therapy prior to each apheresis.
Haemodialysis patients
Anaphylactoid reactions have been reported in patients dialysed with high-flux membranes (e.g., AN 69) and treated concomitantly with an ACE inhibitor. In these patients consideration should be given to using a different type of dialysis membrane or a different class of antihypertensive agent.
Primary aldosteronism
Patients with primary hyperaldosteronism generally will not respond to anti-hypertensive drugs acting through inhibition of the renin-angiotensin system. Therefore, the use of this product is not recommended.
Pregnancy
ACE inhibitors should not be initiated during pregnancy. Unless continued ACE inhibitor therapy is considered essential, patients planning pregnancy should be changed to alternative anti-hypertensive treatments which have an established safety profile for use in pregnancy. When pregnancy is diagnosed, treatment with ACE inhibitors should be stopped immediately, and, if appropriate, alternative therapy should be started (see sections 4.3 and 4.6).
Hepatic encephalopathy
When liver function is impaired, thiazide diuretics and thiazide-related diuretics may cause, particularly in case of electrolyte imbalance, hepatic encephalopathy which can progress to hepatic coma. Administration of the diuretic should be stopped immediately if this occurs.
Photosensitivity
Cases of photosensitivity reactions have been reported with thiazides and related thiazides diuretics (see section 4.8). If photosensitivity reaction occurs during treatment, it is recommended to stop the treatment. If a re-administration of the diuretic is deemed necessary, it is recommended to protect exposed areas to the sun or to artificial UVA.
Precautions for use
Renal function
- In cases of severe renal impairment (creatinine clearance <30 mL/min), treatment is contraindicated.
- For patients with a moderate renal impairment (creatinine clearance <60 mL/min), treatment is contraindicated with Triplixam doses containing 10mg/2.5mg of perindopril/indapamide combination (i.e., Triplixam 10mg/2.5mg /5mg and 10mg/2.5mg/10mg).
- In certain hypertensive patients without pre-existing apparent renal lesions and for whom renal blood tests show functional renal insufficiency, treatment should be stopped and possibly restarted either at a low dose or with one constituent only.
In these patients usual medical follow-up will include frequent monitoring of potassium and creatinine, after two weeks of treatment and then every two months during therapeutic stability period. Renal failure has been reported mainly in patients with severe heart failure or underlying renal failure including renal artery stenosis.
The drug is usually not recommended in case of bilateral renal artery stenosis or a single functioning kidney. - Risk of arterial hypotension and/or renal insufficiency (in cases of cardiac insufficiency , water and electrolyte depletion, etc…): Marked stimulation of the renin-angiotensin-aldosterone system has been observed with perindopril particularly during marked water and electrolyte depletions (strict sodium restricted diet or prolonged diuretic treatment), in patients whose blood pressure was initially low, in cases of renal artery stenosis, congestive heart failure or cirrhosis with oedema and ascites.
The blocking of this system with an angiotensin converting enzyme inhibitor may therefore cause, particularly at the time of the first administration and during the first two weeks of treatment, a sudden drop in blood pressure and/or an increase in plasma levels of creatinine, showing a functional renal insufficiency. Occasionally this can be acute in onset, although rare, and with a variable time to onset.
In such cases, the treatment should then be initiated at a lower dose and increased progressively. In patients with ischaemic heart or cerebrovascular disease an excessive fall in blood pressure could result in a myocardial infarction or cerebrovascular accident. - Thiazide diuretics and thiazide-related diuretics are only fully effective when renal function is normal or only slightly impaired (creatinine levels lower than approximately 25 mg/l, i.e. 220 µmol/l for an adult). In the elderly the value of plasma creatinine levels should be adjusted in relation to age, weight and gender.
Hypovolaemia, secondary to the loss of water and sodium caused by the diuretic at the start of treatment, causes a reduction in glomerular filtration. It may result in an increase in blood urea and creatinine levels. This transitory functional renal insufficiency is of no adverse consequence in patients with normal renal function but may however worsen a pre-existing renal impairment. - Amlodipine may be used at normal doses in patients with renal failure. Changes in amlodipine plasma concentrations are not correlated with degree of renal impairment.
- The effect of the combination Triplixam has not been tested in renal dysfunction. In renal impairment, Triplixam doses should respect those of the individual components taken separately.
Hypotension and water and sodium depletion
- There is a risk of sudden hypotension in the presence of pre-existing sodium depletion (in particular in individuals with renal artery stenosis). Therefore systematic testing should be carried out for clinical signs of water and electrolyte depletion, which may occur with an intercurrent episode of diarrhoea or vomiting. Regular monitoring of plasma electrolytes should be carried out in such patients.
Marked hypotension may require the implementation of an intravenous infusion of isotonic saline. Transient hypotension is not a contraindication to continuation of treatment. After re-establishment of a satisfactory blood volume and blood pressure, treatment can be started again either at a reduced dose or with only one of the constituents. - Reduction in sodium levels can be initially asymptomatic and regular testing is therefore essential. Testing should be more frequent in elderly and cirrhotic patients (see sections 4.8 and 4.9). Any diuretic treatment may cause hyponatraemia, sometimes with very serious consequences. Hyponatraemia with hypovolaemia may be responsible of dehydration and orthostatic hypotension. Concomitant loss of chloride ions may lead to secondary compensatory metabolic alkalosis: the incidence and degree of this effect are slight.
Potassium levels
- The combination of indapamide with perindopril and amlodipine does not prevent the onset of hypokalaemia particularly in diabetic patients or in patients with renal failure. As with any antihypertensive agent in combination with a diuretic, regular monitoring of plasma potassium levels should be carried out.
- Elevations in serum potassium have been observed in some patients treated with ACE inhibitors, including perindopril. ACE inhibitors can cause hyperkalaemia because they inhibit the release of aldosterone. The effect is usually not significant in patients with normal renal function. Risk factors for the development of hyperkalaemia include those with renal insufficiency, worsening of renal function, age (>70 years), diabetes mellitus, intercurrent events, in particular dehydration, acute cardiac decompensation, metabolic acidosis and concomitant use of potassium-sparing diuretics (e.g., spironolactone, eplerenone, triamterene, or amiloride), potassium supplements or potassium-containing salt substitutes; or those patients taking other drugs associated with increases in serum potassium (e.g. heparin, co-trimoxazole also known as trimethoprim/sulfamethoxazole) and especially aldosterone antagonists or angiotensin-receptor blockers. The use of potassium supplements, potassium-sparing diuretics, or potassium-containing salt substitutes particularly in patients with impaired renal function may lead to a significant increase in serum potassium. Hyperkalaemia can cause serious, sometimes fatal arrhythmias. Potassium-sparing diuretics and angiotensin-receptor blockers should be used with caution in patients receiving ACE inhibitors, and serum potassium and renal function should be monitored. If concomitant use of the above-mentioned agents is deemed appropriate, they should be used with caution and with frequent monitoring of serum potassium (see section 4.5).
- Potassium depletion with hypokalaemia is a major risk with thiazide diuretics and thiazide-related diuretics. Hypokalaemia may cause muscle disorders. Cases of rhabdomyolysis have been reported, mainly in the context of severe hypokalaemia. The risk of onset of lowered potassium levels (<3.4 mmol/l) should be prevented in some high risk populations such as elderly and/or malnourished subjects, whether or not they are taking multiple medications, cirrhotic patients with oedema and ascites, coronary patients and patients with heart failure.
In such cases hypokalaemia increases the cardiac toxicity of cardiac glycosides and the risk of rhythm disorders.
Subjects presenting with a long QT interval are also at risk, whether the origin is congenital or iatrogenic. Hypokalaemia, as with bradycardia, acts as a factor which favours the onset of severe rhythm disorders, in particular torsades de pointes, which may be fatal.
In all cases more frequent testing of potassium levels is necessary. The first measurement of plasma potassium levels should be carried out during the first week following the start of treatment.
If low potassium levels are detected, correction is required. Hypokalaemia found in association with low serum magnesium concentration can be refractory to treatment unless serum magnesium is corrected.
Calcium levels
Thiazide diuretics and thiazide-related diuretics may reduce urinary excretion of calcium and cause a mild and transient increase in plasma calcium levels. Markedly raised levels of calcium may be related to undiagnosed hyperparathyroidism. In such cases the treatment should be stopped before investigating the parathyroid function (see section 4.8).
Plasma magnesium
Thiazides and related diuretics including indapamide have been shown to increase the urinary excretion of magnesium, which may result in hypomagnesaemia (see section 4.5 and 4.8).
Renovascular hypertension
The treatment for renovascular hypertension is revascularisation. Nonetheless, angiotensin converting enzyme inhibitors can be beneficial in patients presenting with renovascular hypertension who are awaiting corrective surgery or when such a surgery is not possible.
If Triplixam is prescribed to patients with known or suspected renal artery stenosis, treatment should be started in a hospital setting at a low dose and renal function and potassium levels should be monitored, since some patients have developed a functional renal insufficiency which was reversed when treatment was stopped.
Cough
A dry cough has been reported with the use of angiotensin converting enzyme inhibitors. It is characterised by its persistence and by its disappearance when treatment is withdrawn. An iatrogenic aetiology should be considered in the event of this symptom. If the prescription of an angiotensin converting enzyme inhibitor is still preferred, continuation of treatment may be considered.
Atherosclerosis
The risk of hypotension exists in all patients but particular care should be taken in patients with ischaemic heart disease or cerebral circulatory insufficiency, with treatment being started at a low dose.
Hypertensive crisis
The safety and efficacy of amlodipine in hypertensive crisis has not been established.
Cardiac failure/severe cardiac insufficiency
Patients with heart failure should be treated with caution.
In a long-term, placebo controlled study in patients with severe heart failure (NYHA class III and IV) the reported incidence of pulmonary oedema was higher in the amlodipine treated group than in the placebo group. Calcium channel blockers, including amlodipine, should be used with caution in patients with congestive heart failure, as they may increase the risk of future cardiovascular events and mortality. In patients with severe cardiac insufficiency (grade IV) treatment should be started under medical supervision with a reduced initial dose. Treatment with beta-blockers in hypertensive patients with coronary insufficiency should not be stopped: the ACE inhibitor should be added to the beta-blocker.
Aortic or mitral valve stenosis/hypertrophic cardiomyopathy
ACE inhibitors should be used with caution in patient with an obstruction in the outflow tract of the left ventricle.
Diabetic patients
In patients with insulin dependent diabetes mellitus (spontaneous tendency to increased levels of potassium), treatment should be started under medical supervision with a reduced initial dose.
The glycaemia levels should be closely monitored in diabetic patients previously treated with oral antidiabetic drugs or insulin, namely during the first month of treatment with an ACE inhibitor.
Monitoring of blood glucose is important in diabetic patients, particularly when potassium levels are low.
Ethnic differences
As with other angiotensin converting enzyme inhibitors, perindopril is apparently less effective in lowering blood pressure in black people than in non-blacks, possibly because of a higher prevalence of low-renin states in the black hypertensive population.
Surgery / anaesthesia
Angiotensin converting enzyme inhibitors can cause hypotension in cases of anaesthesia, especially when the anaesthetic administered is an agent with hypotensive potential.
It is therefore recommended that treatment with long-acting angiotensin converting enzyme inhibitors such as perindopril should be discontinued where possible one day before surgery.
Hepatic impairment
Rarely, ACE inhibitors have been associated with a syndrome that starts with cholestatic jaundice and progresses to fulminant hepatic necrosis and (sometimes) death. The mechanism of this syndrome is not understood. Patients receiving ACE inhibitors who develop jaundice or marked elevations of hepatic enzymes should discontinue the ACE inhibitor and receive appropriate medical follow-up (see section 4.8).
The half-life of amlodipine is prolonged and AUC values are higher in patients with impaired liver function; dosage recommendations have not been established. Amlodipine should therefore be initiated at the lower end of the dosing range and caution should be used, both on initial treatment and when increasing the dose. Slow dose titration and careful monitoring may be required in patients with severe hepatic impairment.
The effect of the combination Triplixam has not been tested in hepatic dysfunction. Taking into account the effect of each individual component of this combination, Triplixam is contraindicated in patients with severe hepatic impairment, and caution should be exercised in patients with mild to moderate hepatic impairment.
Uric acid
Tendency to gout attacks may be increased in hyperuricaemic patients.
Elderly
Renal function and potassium levels should be tested before the start of treatment. The initial dose is subsequently adjusted according to blood pressure response, especially in cases of water and electrolyte depletion, in order to avoid sudden onset of hypotension.
In the elderly increase of the dosage of amlodipine should take place with care (see sections 4.2 and 5.2).
Excipients
Level of sodium Triplixam contains less than 1 mmol sodium (23 mg) per tablet, i.e. essentially ‘sodium-free’.
Choroidal effusion, acute myopia and secondary angle-closure glaucoma
Sulfonamide or sulfonamide derivative drugs can cause an idiosyncratic reaction resulting in choroidal effusion with visual field defect, transient myopia and acute angle-closure glaucoma. Symptoms include acute onset of decreased visual acuity or ocular pain and typically occur within hours to weeks of drug initiation. Untreated acute angle-closure glaucoma can lead to permanent vision loss. The primary treatment is to discontinue drug intake as rapidly as possible. Prompt medical or surgical treatments may need to be considered if the intraocular pressure remains uncontrolled. Risk factors for developing acute angle-closure glaucoma may include a history of sulfonamide or penicillin allergy.
Athletes
Athletes should note that this product contains an active substance which may cause a positive reaction in doping tests.
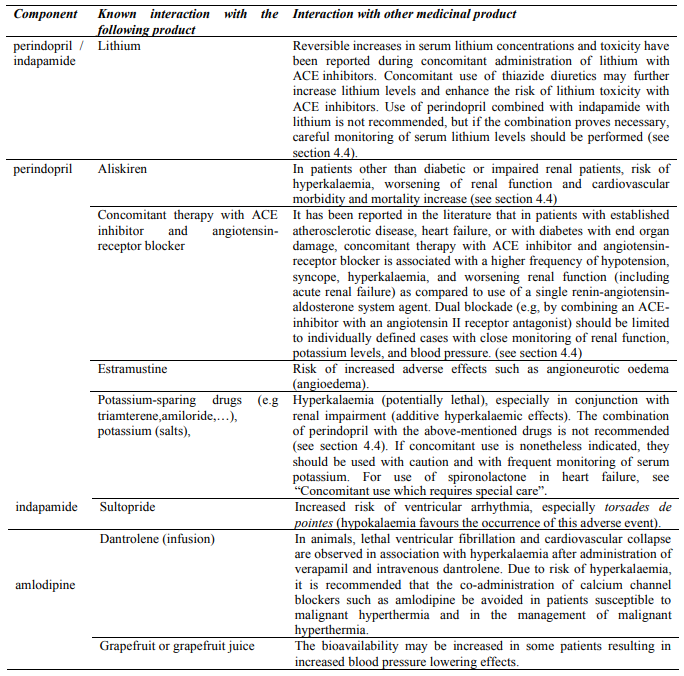
4.5. Interaction with other medicinal products and other forms of interaction
Clinical trial data has shown that dual blockade of the renin-angiotensin-aldosterone-system (RAAS) through the combined use of ACE-inhibitors, angiotensin II receptor blockers or aliskiren is associated with a higher frequency of adverse events such as hypotension, hyperkalaemia and decreased renal function (including acute renal failure) compared to the use of a single RAAS-acting agent (see sections 4.3, 4.4 and 5.1).
Drugs increasing the risk of angioedema
Concomitant use of ACE inhibitors with sacubitril/valsartan is contraindicated as this increases the risk of angioedema (see section 4.3 and 4.4). Sacubitril/valsartan must not be started until 36 hours after taking the last dose of perindopril therapy. Perindopril therapy must not be started until 36 hours after the last dose of sacubitril/valsartan (see sections 4.3 and 4.4).
Concomitant use of ACE inhibitors with racecadotril, mTOR inhibitors (e.g. sirolimus, everolimus, temsirolimus) and gliptins (e.g. linagliptin, saxagliptin, sitagliptin, vildagliptin) may lead to an increased risk for angioedema (see section 4.4).
Drugs inducing hyperkalaemia
Although serum potassium usually remains within normal limits, hyperkalaemia may occur in some patients treated with Triplixam. Some drugs or therapeutic classes may increase the occurrence of hyperkalaemia: aliskiren, potassium salts, potassium-sparing diuretics (e.g. spironolactone, triamterene or amiloride), ACE inhibitors, angiotensin-II receptors antagonists, NSAIDs, heparins, immunosuppressant agents such as ciclosporin or tacrolimus, trimethoprim and cotrimoxazole (trimethoprim/sulfamethoxazole), as trimethoprim is known to act as a potassium-sparing diuretic like amiloride. The combination of these drugs increases the risk of hyperkalaemia. Therefore, the combination of Triplixam with the above-mentioned drugs is not recommended. If concomitant use is indicated, they should be used with caution and with frequent monitoring of serum potassium.
Concomitant use contraindicated (see section 4.3)
Aliskiren: In diabetic or impaired renal patients, risk of hyperkalaemia, worsening of renal function and cardiovascular morbidity and mortality increase.
Extracorporeal treatments : Extracorporeal treatments leading to contact of blood with negatively charged surfaces such as dialysis or haemofiltration with certain high-flux membranes (e.g. polyacrylonitril membranes) and low density lipoprotein apheresis with dextran sulphate due to increased risk of severe anaphylactoid reactions (see section 4.3). If such treatment is required, consideration should be given to using a different type of dialysis membrane or a different class of antihypertensive agent.
Concomitant use not recommended
Concomitant use which requires special care
Concomitant use to be taken into consideration
4.6. Fertility, pregnancy and lactation
Given the effects of the individual components in this combination product on pregnancy and lactation, Triplixam is not recommended during the first trimester of pregnancy. Triplixam is contraindicated during the second and third trimesters of pregnancy.
Triplixam is not recommended during lactation. A decision should therefore be made whether to discontinue nursing or to discontinue Triplixam taking account the importance of this therapy for the mother.
Pregnancy
Perindopril
The use of ACE inhibitors is not recommended during the first trimester of pregnancy (see section 4.4). The use of ACE inhibitors is contra-indicated during the second and third trimesters of pregnancy (see sections 4.3 and 4.4).
Epidemiological evidence regarding the risk of teratogenicity following exposure to ACE inhibitors during the first trimester of pregnancy has not been conclusive; however a small increase in risk cannot be excluded. Unless continued ACE inhibitor therapy is considered essential, patients planning pregnancy should be changed to alternative anti-hypertensive treatments which have an established safety profile for use in pregnancy. When pregnancy is diagnosed, treatment with ACE inhibitors should be stopped immediately, and, if appropriate, alternative therapy should be started.
Exposure to ACE inhibitor therapy during the second and third trimesters is known to induce human foetotoxicity (decreased renal function, oligohydramnios, skull ossification retardation) and neonatal toxicity (renal failure, hypotension, hyperkalaemia) (see section 5.3).
Should exposure to ACE inhibitors have occurred from the second trimester of pregnancy, ultrasound check of renal function and skull is recommended. Infants whose mothers have taken ACE inhibitors should be closely observed for hypotension (see sections 4.3 and 4.4).
Indapamide
There are no or limited amount of data (less than 300 pregnancy outcomes) from the use of indapamide in pregnant women. Prolonged exposure to thiazide during the third trimester of pregnancy can reduce maternal plasma volume as well as uteroplacental blood flow, which may cause a feto-placental ischemia and growth retardation. Moreover, rare cases of hypoglycemia and thrombocytopenia in neonates have been reported following exposure near term.
Amlodipine
The safety of amlodipine in human pregnancy has not been established. In animal studies, reproductive toxicity was observed at high doses (see section 5.3).
Breastfeeding
Triplixam is not recommended during lactation.
Perindopril
Because no information is available regarding the use of perindopril during breastfeeding, perindopril is not recommended and alternative treatments with better established safety profiles during breast-feeding are preferable, especially while nursing a newborn or preterm infant.
Indapamide
There is insufficient information on the excretion of indapamide/metabolites in human milk. Hypersensitivity to sulfonamide-derived medicines and hypokalaemia might occur. A risk to newborns/infants cannot be excluded. Indapamide is closely related to thiazide diuretics which have been associated, during breast-feeding, with a decrease or even suppression of milk lactation.
Amlodipine
Amlodipine is excreted in human milk. The proportion of the maternal dose received by the infant has been estimated with an interquartile range of 3–7%, with a maximum of 15%. The effect of amlodipine on infants is unknown.
Fertility
Common to perindopril and indapamide
Reproductive toxicity studies showed no effect on fertility in female and male rats (see section 5.3). No effects on human fertility are anticipated.
Amlodipine
Reversible biochemical changes in the head of spermatozoa have been reported in some patients treated by calcium channel blockers. Clinical data are insufficient regarding the potential effect of amlodipine on fertility. In one rat study, adverse effects were found on male fertility (see section 5.3).
4.7. Effects on ability to drive and use machines
No studies on the effects of Triplixam on the ability to drive and use machines have been performed. Perindopril and indapamide have no influence on the ability to drive and use machines but individual reactions related to low blood pressure may occur in some patients.
Amlodipine can have minor or moderate influence on the ability to drive and use machines. If patients suffer from dizziness, headache, fatigue, weariness or nausea, the ability to react may be impaired.
As a result the ability to drive or operate machinery may be impaired. Caution is recommended especially at the start of treatment.
4.8. Undesirable effects
Summary of the safety profile
The most commonly reported adverse reactions with perindopril, indapamide and amlodipine given separately are: hypokalaemia, dizziness, headache, paraesthesia, somnolence, dysgeusia, visual impairment, diplopia, tinnitus, vertigo, palpitations, flushing, hypotension (and effects related to hypotension), cough, dyspnoea, gastro-intestinal disorders (abdominal pain, constipation, diarrhoea, dyspepsia, nausea, vomiting, change in bowel habit), pruritus, rash, rash maculo-papular, muscle spasms, ankle swelling, asthenia, oedema and fatigue.
Tabulated list of adverse reactions
The following undesirable effects have been observed with perindopril, indapamide or amlodipine during treatment and ranked under the following frequency: Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1000 to <1/100); rare (≥1/10000 to <1/1000), very rare (<1/10000); not known (cannot be estimated from the available data).
| MedDRA System Organ Class | Undesirable Effects | Frequency | ||
|---|---|---|---|---|
| Perindopril | Indapamide | Amlodipine | ||
| Infections and infestations | Rhinitis | Very rare | - | Uncommon |
| Endocrine disorders | Syndrome of inappropriate antidiuretic hormone secretion (SIADH) | Rare | - | |
| Blood and Lymphatic System Disorders | Eosinophilia | Uncommon* | - | - |
| Agranulocytosis (see section 4.4) | Very rare | Very rare | - | |
| Aplastic anaemia | - | Very rare | ||
| Pancytopenia | Very rare | - | - | |
| Leukopenia (see section 4.4) | Very rare | Very rare | Very rare | |
| Neutropenia (see section 4.4) | Very rare | - | - | |
| Haemolytic anaemia | Very rare | Very rare | - | |
| Thrombocytopenia (see section 4.4) | Very rare | Very rare | Very rare | |
| Immune System Disorders | Hypersensitivity | - | Uncommon | Very rare |
| Metabolism and Nutrition Disorders | Hypokalaemia (see section 4.4) | - | Common | - |
| Hypoglycaemia (see sections 4.4 and 4.5) | Uncommon* | - | - | |
| Hyperkalaemia reversible on discontinuation (see section 4.4) | Uncommon* | - | - | |
| Hyponatraemia (see section 4.4) | Uncommon* | Uncommon | ||
| Hypochloraemia | - | Rare | - | |
| Hypomagnesaemia | - | Rare | - | |
| Hyperglycaemia | - | - | Very rare | |
| Hypercalcaemia | - | Very rare | - | |
| Psychiatric disorders | Insomnia | - | - | Uncommon |
| Mood altered (including anxiety) | Uncommon | - | Uncommon | |
| Depression | Uncommon* | - | Uncommon | |
| Sleep disorder | Uncommon | - | - | |
| Confusional state | Very rare | - | Rare | |
| Nervous System disorders | Dizziness | Common | - | Common |
| Headache | Common | Rare | Common | |
| Paraesthesia | Common | Rare | Uncommon | |
| Somnolence | Uncommon* | - | Common | |
| Hypoaesthesia | - | - | Uncommon | |
| Dysgeusia | Common | - | Uncommon | |
| Tremor | - | - | Uncommon | |
| Syncope | Uncommon* | Not known | Uncommon | |
| Hypertonia | - | - | Very rare | |
| Neuropathy peripheral | - | - | Very rare | |
| Extrapyramidal disorder (extrapyramidal syndrome) | - | - | Not known | |
| Stroke possibly secondary to excessive hypotension in high-risk patients (see section 4.4) | Very rare | - | - | |
| Possibility of onset of hepatic encephalopathy in case of hepatic insufficiency (see sections 4.3 and 4.4) | - | Not known | - | |
| Eye Disorders | Visual impairment | Common | Not known | Common |
| Acute angle-closure glaucoma | - | Not known | - | |
| Choroidal effusion | - | Not known | - | |
| Diplopia | - | - | Common | |
| Myopia | - | Not known | - | |
| Vision blurred | - | Not known | - | |
| Ear and labyrinth disorders | Tinnitus | Common | - | Uncommon |
| Vertigo | Common | Rare | - | |
| Cardiac Disorders | Palpitations | Uncommon* | - | Common |
| Tachycardia | Uncommon* | - | - | |
| Angina pectoris (see section 4.4) | Very rare | - | - | |
| Arrhythmia (including bradycardia, ventricular tachycardia and atrial fibrillation) | Very rare | Very rare | Uncommon | |
| Myocardial infarction, possibly secondary to excessive hypotension in high risk patients (see section 4.4) | Very rare | - | Very rare | |
| Torsade de pointes (potentially fatal) (see sections 4.4 and 4.5) | - | Not known | - | |
| Vascular Disorders | Flushing | Rare* | - | Common |
| Hypotension (and effects related to hypotension) (see section 4.4) | Common | Very rare | Uncommon | |
| Vasculitis | Uncommon* | - | Very rare | |
| Raynaud’s phenomenon | Not known | - | - | |
| Respiratory, Thoracic and Mediastinal Disorders | Cough (see section 4.4) | Common | - | Uncommon |
| Dyspnoea | Common | - | Common | |
| Bronchospasm | Uncommon | - | - | |
| Eosinophilic pneumonia | Very rare | - | - | |
| Gastro-intestinal Disorders | Abdominal pain | Common | - | Common |
| Constipation | Common | Rare | Common | |
| Diarrhoea | Common | - | Common | |
| Dyspepsia | Common | - | Common | |
| Nausea | Common | Rare | Common | |
| Vomiting | Common | Uncommon | Uncommon | |
| Dry mouth | Uncommon | Rare | Uncommon | |
| Change of bowel habit | - | - | Common | |
| Gingival hyperplasia | - | - | Very rare | |
| Pancreatitis | Very rare | Very rare | Very rare | |
| Gastritis | - | - | Very rare | |
| Hepato-biliaryvDisorders | Hepatitis (see section 4.4) | Very rare | Not known | Very rare |
| Jaundice | - | - | Very rare | |
| Hepatic function abnormal | - | Very rare | - | |
| Skin and Subcutaneous Tissue Disorders | Pruritus | Common | - | Uncommon |
| Rash | Common | - | Uncommon | |
| Rash maculo-papular | Common | - | - | |
| Urticaria (see section 4.4) | Uncommon | Very rare | Uncommon | |
| Angioedema (see section 4.4) | Uncommon | Very rare | Very rare | |
| Alopecia | - | - | Uncommon | |
| Purpura | - | Uncommon | Uncommon | |
| Skin discolouration | - | - | Uncommon | |
| Hyperhidrosis | Uncommon | - | Uncommon | |
| Exanthema | - | - | Uncommon | |
| Photosensitivity reaction | Uncommon* | Not known (see section 4.4) | Very rare | |
| Psoriasis aggravation | Rare | - | - | |
| Pemphigoid | Uncommon* | - | - | |
| Erythema multiforme | Very rare | - | Very rare | |
| Stevens-Johnson Syndrome | - | Very rare | Very rare | |
| Exfoliative dermatitis | - | - | Very rare | |
| Toxic epidermal necrolysis | - | Very rare | Not known | |
| Quincke’s oedema | - | - | Very rare | |
| Musculoskeletal And Connective Tissue Disorders | Muscle spasms | Common | Not known | Common |
| Ankle swelling | - | - | Common | |
| Arthralgia | Uncommon* | - | Uncommon | |
| Muscular weakness | - | Not known | - | |
| Myalgia | Uncommon* | Not known | Uncommon | |
| Rhabdomyolysis | - | Not known | - | |
| Back pain | - | - | Uncommon | |
| Possible worsening of pre-existing systemic lupus erythematosus | - | Not known | - | |
| Renal and Urinary Disorders | Micturition disorder | - | - | Uncommon |
| Nocturia | - | - | Uncommon | |
| Pollakiuria | - | - | Uncommon | |
| Anuria/oliguria | Rare* | - | - | |
| Acute renal failure | Rare | - | - | |
| Renal failure | Uncommon | Very rare | - | |
| Reproductive System and Breast Disorders | Erectile dysfunction | Uncommon | Uncommon | Uncommon |
| Gynaecomastia | - | - | Uncommon | |
| General Disorders and Administration Site Conditions | Asthenia | Common | - | Common |
| Fatigue | - | Rare | Common | |
| Oedema | - | - | Very common | |
| Chest pain | Uncommon* | - | Uncommon | |
| Pain | - | - | Uncommon | |
| Malaise | Uncommon* | - | Uncommon | |
| Oedema peripheral | Uncommon* | - | - | |
| Pyrexia | Uncommon* | - | - | |
| Investigations | Weight increased | - | - | Uncommon |
| Weight decreased | - | - | Uncommon | |
| Blood urea increased | Uncommon* | - | - | |
| Blood creatinine increased | Uncommon* | - | - | |
| Blood bilirubin increased | Rare | - | - | |
| Hepatic enzyme increased | Rare | Not known | Very rare | |
| Haemoglobin decreased and haematocrit decreased (see section 4.4) | Very rare | - | - | |
| Electrocardiogram QT prolonged (see sections 4.4 and 4.5) | - | Not known | - | |
| Blood glucose increased | - | Not known | - | |
| Blood uric acid increased | - | Not known | - | |
| Injury, poisoning and procedural complications | Fall | Uncommon* | - | - |
* Frequency calculated from clinical trials for adverse events detected from spontaneous report
Description of selected adverse reactions
During phase II and III studies comparing indapamide 1.5mg and 2.5mg, plasma potassium analysis showed a dose-dependent effect of indapamide:
- Indapamide 1.5mg: Plasma potassium <3.4 mmol/l was seen in 10% of patients and <3.2 mmol/l in 4% of patients after 4 to 6 weeks treatment. After 12 weeks treatment, the mean fall in plasma potassium was 0.23 mmol/l.
- Indapamide 2.5 mg: Plasma potassium <3.4 mmol/l was seen in 25% of patients and <3.2 mmol/l in 10% of patients after 4 to 6 weeks treatment. After 12 weeks treatment, the mean fall in plasma potassium was 0.41 mmol/l.
Reporting of suspected adverse reactions
Reporting suspected adverse reactions after authorisation of the medicinal product is important. It allows continued monitoring of the benefit/risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions via the national reporting system.
6.2. Incompatibilities
Not applicable.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.