ZELDOX Capsule Ref.[27776] Active ingredients: Ziprasidone
Source: Health Products and Food Branch (CA) Revision Year: 2020
Action and clinical pharmacology
ZELDOX (ziprasidone hydrochloride) is an atypical antipsychotic agent for oral administration.
Mechanism of Action
The mechanism of action of ziprasidone, as with other drugs having efficacy in schizophrenia, is unknown. However, it has been proposed that the efficacy of this drug in schizophrenia is mediated through a combination of dopamine type 2 (D2) and serotonin type 2 (5HT2) antagonism. Antagonism at receptors other than dopamine and 5HT2 with similar receptor affinities may explain some of the other therapeutic and side effects of ziprasidone. Antagonism of histamine H1 receptors may explain the somnolence observed with ziprasidone. Antagonism of α1-adrenergic receptors may explain the orthostatic hypotension observed with ziprasidone.
Pharmacodynamics
Ziprasidone exhibited high in vitro binding affinity for the dopamine D2 and D3, the serotonin 5-HT2A, 5- HT2C, 5-HT1A, 5-HT1D, and α1-adrenergic receptors (Ki = 4.8, 7.2, 0.4, 1.3, 3.4, 2, and 10 nM, respectively), and moderate affinity for the histamine H1 receptor (Ki=47 nM). Ziprasidone functioned as an antagonist at the D2, 5-HT2A, and 5-HT1D receptors, and as an agonist at the 5-HT1A receptor. Ziprasidone inhibited synaptic reuptake of serotonin and norepinephrine. No appreciable affinity was exhibited for other receptor/binding sites tested, including the cholinergic muscarinic receptor (IC50 >1 µM).
Pharmacokinetics
Overview
The multiple-dose pharmacokinetics of ziprasidone are dose-proportional within the proposed clinical dose range, and ziprasidone accumulation is predictable with multiple dosing. Steady–state is attained within 1-3 days when dosing as recommended. The mean terminal phase half-life after multiple dosing in normal volunteers and schizophrenic patients is between 6 and 10 hours, with a range of individual values from 3 to 18 hours.
Ziprasidone's activity is primarily due to the parent drug. Ziprasidone is extensively metabolized after oral administration with only a small amount excreted in the urine (<1%) or feces (<4%) as unchanged drug. Ziprasidone is unlikely to interfere with the metabolism of drugs metabolized by cytochrome P450 enzymes.
Absorption
Following oral administration of multiple doses of ziprasidone with food, peak serum concentrations typically occur 6 to 8 hours post-dose. The absolute bioavailability of a 20 mg dose under fed conditions is approximately 60%. The absorption of ziprasidone is increased up to two-fold in the presence of food.
Distribution
Ziprasidone has a mean apparent volume of distribution of 1.5 L/kg. Twice daily dosing generally leads to attainment of steady state within 1–3 days.
Ziprasidone is greater than 99% bound to plasma proteins, binding primarily to albumin and α1-acid glycoprotein. The in vitro plasma protein binding of ziprasidone was not altered by warfarin or propranolol, two highly protein-bound drugs, nor did ziprasidone alter the binding of these drugs in human plasma. Thus, the potential for drug interactions with ziprasidone due to displacement is minimal.
Metabolism
Ziprasidone is extensively metabolized after oral administration with only a small amount excreted as unchanged drug in the urine (<1%) or feces (<4%). Unchanged ziprasidone represents about 44% of total drug-related material in serum.
Ziprasidone is primarily cleared via three metabolic routes (one route begins with reduction, the other two with oxidation) to yield four major circulating metabolites: S-methyldihydroziprasidone, via reduction then methylation, and benzisothiazolepiperazine (BITP) sulphoxide, BITP-sulphone, and ziprasidone sulphoxide via oxidation routes
Based on in vivo abundance of excretory metabolites, approximately two-thirds of ziprasidone metabolic clearance is mediated via reduction and methylation to generate S-methyldihydroziprasidone, while cytochrome P450-catalyzed oxidation mediates less than one third of ziprasidone clearance
In vitro studies using human liver subcellular fractions indicate that the metabolite S-methyldihydroziprasidone is generated in two steps: the reduction reaction is mediated by aldehyde oxidase and potentially also by glutathione, while the subsequent methylation is mediated by thiol methyltransferase. There are no known clinically relevant inhibitors or inducers of aldehyde oxidase. In vitro studies using human liver microsomes and recombinant enzymes indicate that CYP3A4 is the major CYP contributing to the two routes of oxidative metabolism of ziprasidone. CYP1A2 may contribute to a much lesser extent.
Excretion
The mean terminal phase half-life after multiple dosing in normal volunteers and schizophrenic patients is between 6 and 10 hours, with a range of individual values from 3 to 18 hours. Approximately 20% of ziprasidone dose is excreted in the urine, with approximately 66% being eliminated in the feces. S-methyldihydroziprasidone is mainly eliminated by biliary excretion and CYP3A4 metabolism. The sulphoxide is eliminated through renal excretion and by secondary metabolism catalyzed by CYP3A4.
Special Populations and Conditions
Pediatrics
Safety and efficacy of ZELDOX in children have not been established.
Age and Gender
In a multiple-dose (8 days of treatment) study involving n=32 subjects, there was no difference in the pharmacokinetics of ziprasidone between men and women or between elderly (>65 years) and young (18 to 45 years) subjects. Additionally, population pharmacokinetic evaluation of patients in controlled trials has revealed no evidence of clinically significant age or gender-related differences in the pharmacokinetics of ziprasidone. Dosage modifications for age or gender are, therefore, not recommended.
Race
No specific pharmacokinetic study was conducted to investigate the effects of race. Population pharmacokinetic evaluation has revealed no evidence of clinically significant race-related differences in the pharmacokinetics of ziprasidone. Dosage modifications for race are, therefore, not recommended.
Hepatic Insufficiency
As ziprasidone is cleared substantially by the liver, the presence of hepatic impairment would be expected to increase the AUC of ziprasidone; a multiple-dose study at the lowest therapeutic dose of 20 mg BID for 5 days in subjects with clinically significant (Childs-Pugh Class A and B) cirrhosis (n=13) revealed an increase in AUC0-12 of 19% and 34% respectively, compared to a matched control group (n=13). A halflife of 7.1 hours was observed in subjects with cirrhosis compared to 4.8 hours in the control group. The effect of liver impairment on the serum concentrations of the metabolites is unknown.
Renal Insufficiency
Because ziprasidone is highly metabolized with less than 1% of the drug excreted unchanged, renal impairment alone is unlikely to have a major impact on the pharmacokinetics of ziprasidone. The pharmacokinetic characteristics of ziprasidone following 8 days of treatment with 20 mg BID were similar among subjects with varying degrees of renal impairment (n=27), and subjects with normal renal function (n=9), indicating that dosage adjustment based upon the degree of renal impairment is not required. Ziprasidone is not removed by hemodialysis.
Detailed pharmacology
ANIMAL
Pharmacodynamics
Ziprasidone exhibits potent effects in preclinical assays predictive of antipsychotic activity. While the compound was found to be a dopamine antagonist in vitro and in vivo, its most potent action is antagonism of serotonin 5-HT2A receptors, where its affinity was an order of magnitude greater than that observed for dopamine D2 receptor sites. In vivo, ziprasidone antagonized 5-HT2A receptor agonistinduced head twitch with six-fold higher potency than was required to block d-amphetamine-induced hyperactivity, a measure of central D2 receptor antagonism which is predictive of antipsychotic efficacy. Ziprasidone also had high affinity for the 5-HT1A (agonist), 5-HT1D (antagonist), and 5-HT2C (antagonist) serotonin receptor subtypes, and blocked the neuronal reuptake of norepinephrine and serotonin with moderate affinity. Ziprasidone was found to enhance the release of dopamine in rat prefrontal cortex.
The potential for antipsychotic efficacy without severe motor side effects is supported by the relatively weak potency of ziprasidone to produce catalepsy in animals, contrasted with its potent antagonism of conditioned avoidance responding and dopamine receptor agonist-induced locomotor activation and stereotypy.
In addition to the animal studies of antipsychotic efficacy and mechanism of action, a general pharmacological evaluation of ziprasidone was conducted to obtain a more extensive characterization of its actions on various organ systems in vitro and in vivo. In general, ziprasidone was well tolerated in animals at doses that produced effective dopamine receptor antagonism in the brain. Cardiovascular changes in dog were limited to mild increases in heart rate after oral doses of ziprasidone that achieved 2- to 4-fold higher plasma levels than the plasma Cmax associated with the maximum recommended human dose.
Respiratory function, as judged by blood gas measurements, gastrointestinal motility and renal function (24-hour), were not perturbed by effective dopamine receptor antagonist doses of ziprasidone. Like other D2 receptor antagonists, ziprasidone did not appear to act as a potent inhibitor of gastric acid secretion in pylorus ligated rats. In vitro, ziprasidone antagonized both α1-adrenoceptor and histamine H1 receptorinduced contractions in isolated guinea pig aorta and ileum, respectively. These effects occurred at concentrations at least 8-fold higher than ziprasidone's Ki for antagonizing D2 receptors in vitro. Ziprasidone had no effects on isolated uterine smooth muscle in rat or on histamine-induced chronotropic activity in guinea pig atrial strips.
Pharmacokinetics
Oral bioavailability was generally less than 40% in mice, rats and dogs, and 60% in humans. The low oral bioavailability in animals was due to incomplete absorption of the dose as indicated by the >50% recovery of the dose in feces as unchanged drug in mice, rats and dogs administered a radiolabeled dose.
There was a 2.5- to 10-fold difference in the half-life observed in mice and rats as compared to that observed in dogs and man. This difference in half-life between rodents and non-rodent species was due to the larger volume of distribution observed in the dogs, and the lower clearance observed in both dogs and humans. In rats and dogs administered multiple doses of ziprasidone, drug exposure at the end of the study was similar to drug exposure at the start of study. Thus, there was no evidence of accumulation and/or metabolic autoinduction after multiple dosing of ziprasidone.
The serum protein binding of ziprasidone is >99% in humans, Long-Evans rats, Sprague-Dawley rats, New Zealand White rabbits and beagle dogs, and greater than 95% in CD-1 mice. In the reproductive toxicity studies in rats and rabbits administered ziprasidone, placental transfer of drug was observed. Studies in pigmented and non-pigmented rats demonstrated that the fractional retention of drug-related material in the eye was due to melanin binding (reversible).
All the metabolites observed in the excreta collected from humans were also observed in the excreta collected from mice, rats and dogs, the species in which the safety studies were conducted. In dogs, rats, mice, and humans, the percentage of circulating radioactivity identified as metabolites was approximately 83%, 50%, 81% and 54%, respectively. Ziprasidone-sulfoxide and -sulfone were the major metabolites in serum collected from all species including humans.
HUMAN
Pharmacodynamics
In normal volunteer PET studies, serum concentrations between 20 and 40 ng/mL are associated with greater than 65% D2 receptor occupancy and greater than 80% 5-HT2 receptor occupancy.
As with other drugs that antagonize D2 receptors, ziprasidone elevates prolactin levels with acute administration. In normal male volunteers, ziprasidone concentrations correlate with increasing prolactin levels. At steady-state, the magnitude of the response was decreased compared with single dose administration, and returned to baseline levels within 12 hours of dosing.
Prolactin elevations observed in both sexes are transient and minimal. These elevations are not generally sustained during chronic administration.
Pharmacokinetics
Following the administration of multiple doses of ziprasidone under fed conditions, peak serum concentrations typically occur 6 to 8 hours post-dose with steady-state attained within 1 to 3 days. Ziprasidone displays linear kinetics over the clinical dose range. Its half-life ranges from 2.9 to 18.0 hours (5th to 95th percentile; mean of 6.6 hours), and apparent systemic clearance ranges from 3.4 to 13.9 mL/min/kg (5th to 95th percentile; mean 7.5 mL/min/kg). The absolute bioavailability of a 20 mg dose under fed conditions is approximately 60%. The absorption of single oral doses of ziprasidone is increased by 100% in the presence of food. Serum concentrations and half-life do not significantly vary between individuals on the basis of gender, age, renal or hepatic status. Ziprasidone has a volume of distribution of approximately 1.5 L/kg. It is greater than 99% bound to plasma proteins, binding primarily to albumin and α1-acid glycoprotein.
Metabolism and Elimination
Following a single oral dose of 14C/3 H-labeled ziprasidone, only a small amount (<1%) was excreted unchanged in the urine. Approximately 20% of the dose is excreted in the urine, with approximately 66% being eliminated in the feces. Unchanged ziprasidone represents about 44% of total drug-related material in serum.
Based on in vivo abundance of excreted metabolites, approximately two-thirds of ziprasidone is metabolized via reduction by aldehyde oxidase. There are no known clinically relevant inhibitors or inducers of aldehyde oxidase. Less than one-third of ziprasidone metabolic clearance is mediated by cytochrome P450 catalyzed oxidation.
In vitro studies using human liver microsomes and recombinant enzymes indicate that CYP3A4 is the major CYP contributing to the oxidative metabolism of ziprasidone. CYP1A2 may contribute to a much lesser extent.
Effect on Cytochrome P450
In vitro studies utilizing human liver microsomes showed that ziprasidone has little potential to inhibit CYP1A2, CYP2C9, CYP2C19, CYP2D6 and CYP3A4. Thus, ziprasidone is unlikely to cause clinically important drug interactions mediated by these enzymes. Consistent with these in vitro results, studies in normal healthy volunteers showed that ziprasidone did not alter the metabolism of dextromethorphan, a CYP2D6 model substrate, to its major metabolite, dextrorphan, and did not alter the pharmacokinetics of ethinyl estradiol, a CYP3A4 substrate.
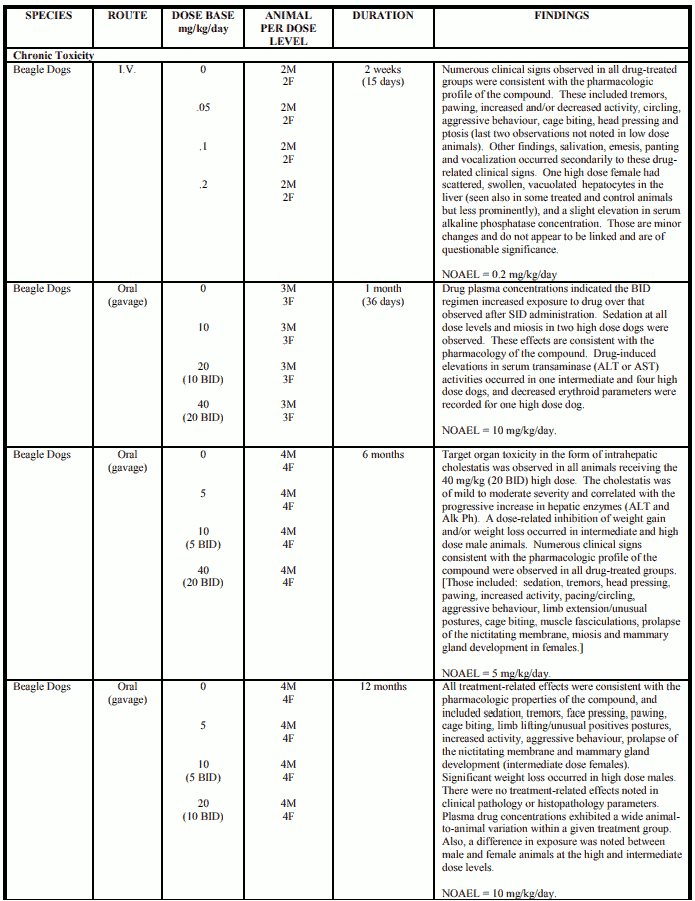
Toxicology
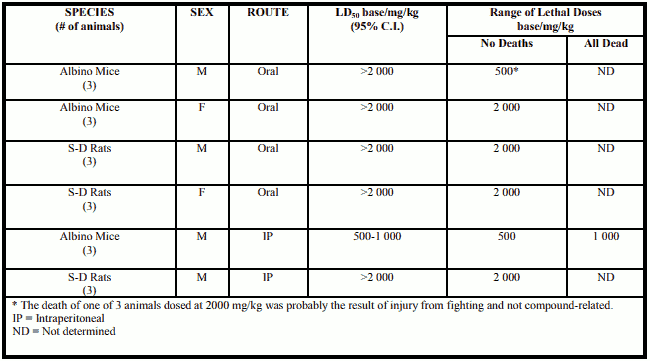
Acute Toxicity - Mice and rats
Description of findings
CP-88,059-1 has a low order of acute toxicity in mice and rats when given either orally or intraperitoneally. No definitive target organs of toxicity were identified, however, clinical signs indicative of CNS effects were produced (especially sedation). Clinical signs included decreased activity and respiration, ptosis and ataxia. Generally within one hour of dosing, the animals became weak, assumed a stationary, prone position, and were barely able to move. Their respiration often became shallow, and several animals were prostrate or nearly prostrate.
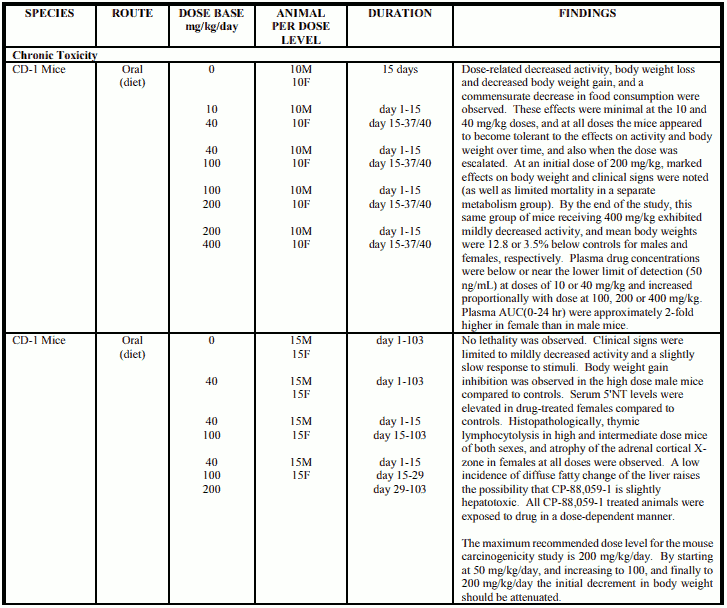
Carcinogenicity
Mice
CP-88,059-1 was administered in the diet of CD-1 mice (50/sex/dose) at an initial dose of 50 mg/kg for the three treated groups. Two groups of 50/sex control mice received unsupplemented feed. On Day 15, the mid and high doses were increased to 100 mg/kg, and on Day 29 the high dose was increased to 200 mg/kg. The final dose levels were therefore 50, 100 and 200 mg/kg. It was concluded, at the end of this 24-month study, that treatment at the mid and high doses produced a statistically significant reduction both in body weight gain during the growth phase of the animals and in body weight in mice at the end of the study compared to controls. This was associated with a reduction in food and water consumption. Histopathological findings were limited to females and consisted of a dose-related increase in the incidence of hyperplasia and neoplasia in the pituitary gland (shown immunohistochemically to be prolactin-producing) and secondary changes in the mammary gland, ovaries and uterus. These findings were seen at 50 to 200 mg/kg/day, corresponding to systemic exposure about 1-4 times greater than that in humans; a no-effect dose level for these effects was not established.
Proliferative changes in the pituitary and mammary glands are not unexpected findings in rodents following treatment with this class of compounds, and are associated with increased prolactin concentrations.
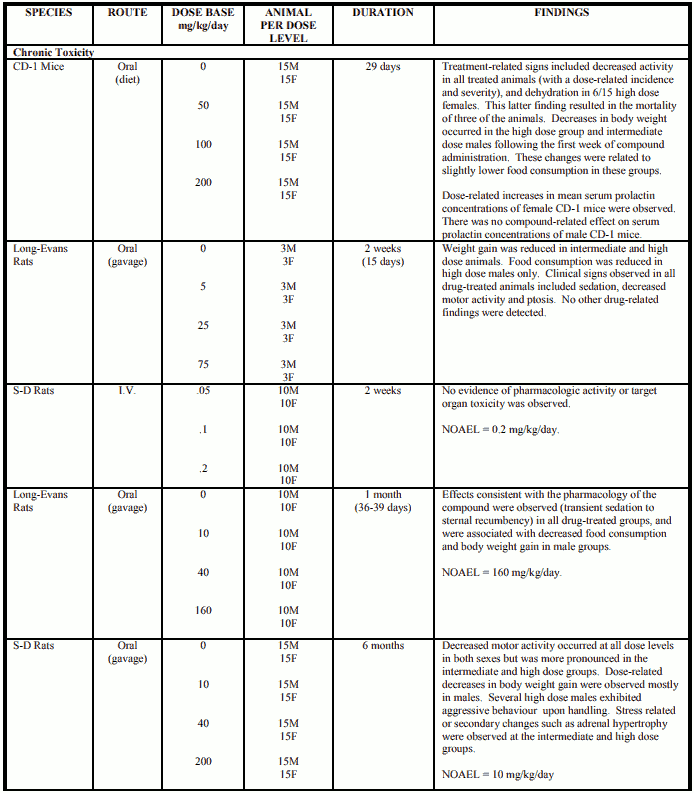
Rats
CP-88,059-1 was administered in the diet of Long-Evans rats (50/sex/level) for 2 years at dose levels of 2, 6 and 12 mg/kg/day. All groups (low, intermediate and high) began at 2 mg/kg/day, and after 2 weeks the intermediate and high dose groups were raised to 6 mg/kg/day. After another two weeks, the high dose level group was increased to 12 mg/kg/day. Two identical control groups (50/sex/group) received non-medicated diet. At dose levels up to 12 mg/kg/day, causing body weight decrements of approximately 10 to 20% relative to controls, ziprasidone showed no oncogenic potential in the rat.
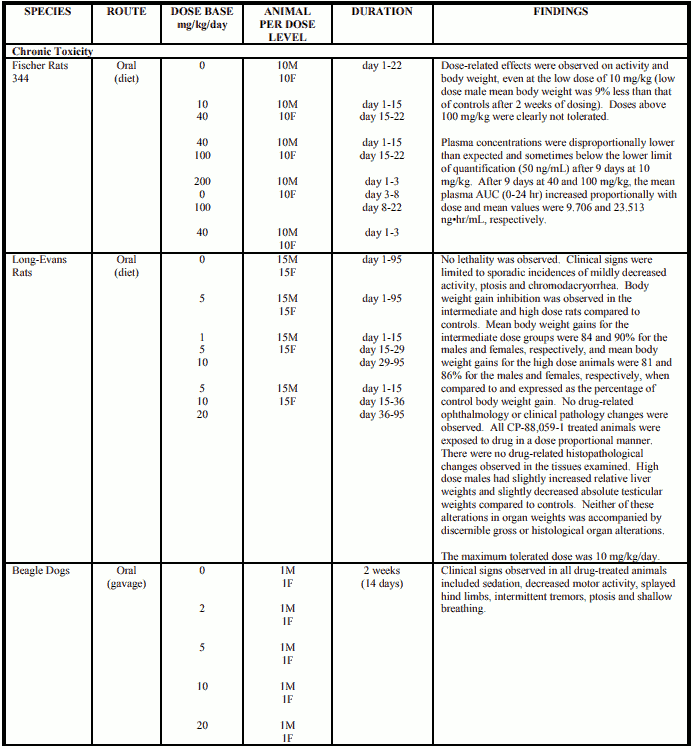
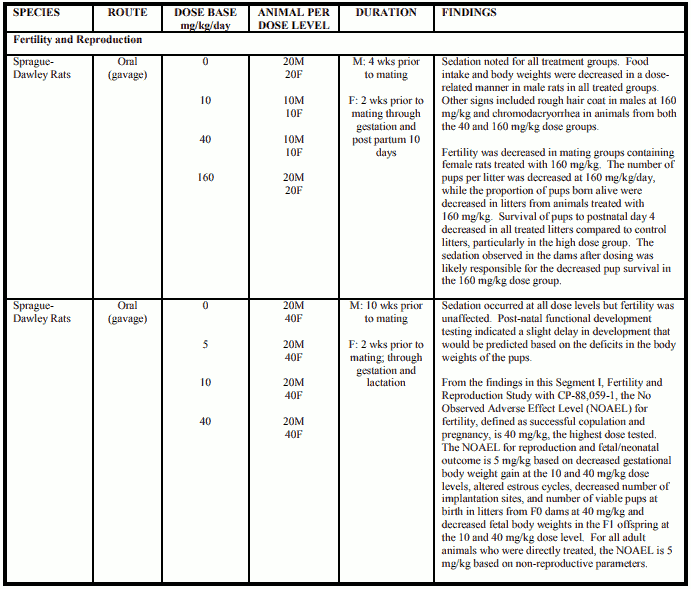
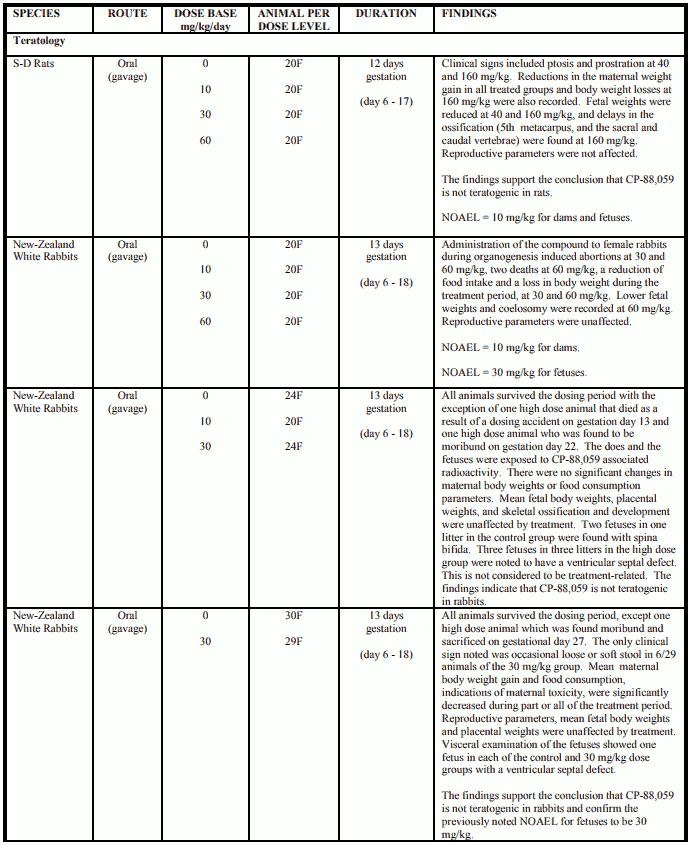
Reproduction and Teratology
Other Studies
Antigenicity Study in Guinea Pigs
CP-88,059-1 does not induce either a systemic anaphylaxis reaction or passive cutaneous anaphylaxis reaction in guinea pigs.
Dermal Toxicity and Ocular Irritation in Rabbits
CP-88,059 is not a Class B Poison or a harmful substance upon either oral or dermal exposure. It is not considered a corrosive material, and it is not an ocular irritant.
Oral Toxicity (Rats), Dermal Toxicity (Rabbits) and Ocular Irritation (Rabbits)
CP-88,059 is not a Class B Poison or a harmful substance upon either oral or dermal exposure. It is not considered a corrosive material, and it is not an ocular irritant.
Acute Phototoxicity (BALB/c Mice)
Ziprasidone did not produce a phototoxic reaction in BALB/c mice as evidenced by the lack of erythema, visible edema, and a statistically significant increase in ear thickness.
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.