FINZALA Chewable tablet, Tablet Ref.[109375] Active ingredients: 17 alpha-Ethinylestradiol Noradrenaline
Source: FDA, National Drug Code (US) Revision Year: 2023
4. Contraindications
Finzala is contraindicated in females who are known to have or develop the following conditions:
- A high risk of arterial or venous thrombotic diseases. Examples include women who are known to:
- Smoke, if over age 35 [see Boxed Warning and Warnings and Precautions (5.1)]
- Have deep vein thrombosis or pulmonary embolism, now or in the past [see Warnings and Precautions (5.1)]
- Have cerebrovascular disease [see Warnings and Precautions (5.1)]
- Have coronary artery disease [see Warnings and Precautions (5.1)]
- Have thrombogenic valvular or thrombogenic rhythm diseases of the heart (for example, subacute bacterial endocarditis with valvular disease, or atrial fibrillation) [see Warnings and Precautions (5.1)]
- Have inherited or acquired hypercoagulopathies [see Warnings and Precautions (5.1)]
- Have uncontrolled hypertension [see Warnings and Precautions (5.4)]
- Have diabetes mellitus with vascular disease [see Warnings and Precautions (5.5)]
- Have headaches with focal neurological symptoms or have migraine headaches with aura
- All women over age 35 with migraine headache [see Warnings and Precautions (5.6)]
- Liver tumors, benign or malignant, or liver disease [see Warnings and Precautions (5.2)]
- Undiagnosed abnormal uterine bleeding [see Warnings and Precautions (5.7)]
- Current diagnosis of, or history of, breast cancer, which may be hormone-sensitive [see Warnings and Precautions (5.10)]
- Use of Hepatitis C drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to the potential for ALT elevations [see Warnings and Precautions (5.3)]
5. Warnings and Precautions
5.1 Thromboembolic Disorders and Other Vascular Problems
Stop Finzala if an arterial or deep venous thrombotic event (VTE) occurs. Stop Finzala if there is unexplained loss of vision, proptosis, diplopia, papilledema, or retinal vascular lesions. Evaluate for retinal vein thrombosis immediately.
If feasible, stop Finzala at least 4 weeks before and through 2 weeks after major surgery or other surgeries known to have an elevated risk of VTE.
Start Finzala no earlier than 4 weeks after delivery, in women who are not breastfeeding. The risk of postpartum VTE decreases after the third postpartum week, whereas the risk of ovulation increases after the third postpartum week.
The use of COCs increases the risk of VTE. However, pregnancy increases the risk of VTE as much or more than the use of COCs. The risk of VTE in women using COCs is 3 to 9 per 10,000 woman-years. The risk of VTE is highest during the first year of use of a COC. The risk of thromboembolic disease due to oral contraceptives gradually disappears after COC use is discontinued.
Use of COCs also increases the risk of arterial thromboses such as strokes and myocardial infarctions, especially in women with other risk factors for these events. COCs have been shown to increase both the relative and attributable risks of cerebrovascular events (thrombotic and hemorrhagic strokes), although, in general, the risk is greatest in older (greater than 35 years of age), hypertensive women who also smoke. COCs also increase the risk for stroke in women with underlying risk factors.
Use COCs with caution in women with cardiovascular disease risk factors.
5.2 Liver Disease
Impaired Liver Function
Do not use Finzala in women with acute viral hepatitis or severe (decompensated) cirrhosis of the liver [see Contraindications (4)]. Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal and COC causation has been excluded. Discontinue Finzala if jaundice develops.
Liver Tumors
Finzala is contraindicated in women with benign and malignant liver tumors [see Contraindications (4)]. Hepatic adenomas are associated with COC use. An estimate of the attributable risk is 3.3 cases per 100,000 COC users. Rupture of hepatic adenomas may cause death through intra-abdominal hemorrhage.
Studies have shown an increased risk of developing hepatocellular carcinoma in long-term (greater than 8 years) COC users. However, the attributable risk of liver cancers in COC users is less than one case per million users.
5.3 Risk of Liver Enzyme Elevations with Concomitant Hepatitis C Treatment
During clinical trials with the Hepatitis C combination drug regimen that contains ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN), including some cases greater than 20 times the ULN, were significantly more frequent in women using ethinyl estradiol-containing medications, such as COCs. Discontinue Finzala prior to starting therapy with the combination drug regimen ombitasvir/paritaprevir/ritonavir, with or without dasabuvir [see Contraindications (4)]. Finzala can be restarted approximately 2 weeks following completion of treatment with the Hepatitis C combination drug regimen.
5.4 High Blood Pressure
Finzala is contraindicated in women with uncontrolled hypertension or hypertension with vascular disease [see Contraindications (4)]. For women with well-controlled hypertension, monitor blood pressure and stop Finzala if blood pressure rises significantly.
An increase in blood pressure has been reported in women taking COCs, and this increase is more likely in older women with extended duration of use. The incidence of hypertension increases with increasing concentrations of progestin.
5.5 Gallbladder Disease
Studies suggest a small increased relative risk of developing gallbladder disease among COC users. Use of COCs may also worsen existing gallbladder disease.
A past history of COC-related cholestasis predicts an increased risk with subsequent COC use. Women with a history of pregnancy-related cholestasis may be at an increased risk for COC-related cholestasis.
5.6 Carbohydrate and Lipid Metabolic Effects
Carefully monitor prediabetic and diabetic women who are taking Finzala. COCs may decrease glucose tolerance in a dose-related fashion.
Consider alternative contraception for women with uncontrolled dyslipidemias. A small proportion of women will have adverse lipid changes while on COCs.
Women with hypertriglyceridemia, or a family history thereof, may be at an increased risk of pancreatitis when using COCs.
5.7 Headache
If a woman taking Finzala develops new headaches that are recurrent, persistent, or severe, evaluate the cause and discontinue Finzala if indicated.
Consider discontinuation of Finzala in the case of increased frequency or severity of migraine during COC use (which may be prodromal of a cerebrovascular event) [see Contraindications (4)].
5.8 Bleeding Irregularities and Amenorrhea
Unscheduled Bleeding and Spotting
Unscheduled (breakthrough or intracyclic) bleeding and spotting sometimes occur in patients on COCs, especially during the first three months of use. If bleeding persists or occurs after previously regular cycles, check for causes such as pregnancy or malignancy. If pathology and pregnancy are excluded, bleeding irregularities may resolve over time or with a change to a different COC.
Based on patient diaries from a clinical trial evaluating the safety and efficacy of a 24-day regimen of norethindrone acetate 1 mg/ethinyl estradiol 20 mcg tablets, 24% to 35% of women experienced unscheduled bleeding per cycle. A total of 10 subjects out of 743 (1.3%) discontinued due to bleeding or spotting.
Amenorrhea and Oligomenorrhea
Women who are not pregnant and use Finzala may experience amenorrhea. In the clinical trial with a 24-day regimen of norethindrone acetate 1 mg/ethinyl estradiol 20 mcg tablets and ferrous fumarate tablets, 22% to 36% of the women using norethindrone acetate 1 mg/ethinyl estradiol 20 mcg tablets and ferrous fumarate tablets experienced amenorrhea in at least one of 6 cycles of use. Some women may experience post-pill amenorrhea or oligomenorrhea, especially when such a condition was preexistent.
If scheduled (withdrawal) bleeding does not occur, consider the possibility of pregnancy. If the patient has not adhered to the prescribed dosing schedule (missed one or more active tablets or started taking them on a day later than she should have), consider the possibility of pregnancy at the time of the first missed period and take appropriate diagnostic measures. If the patient has adhered to the prescribed regimen and misses two consecutive periods, rule out pregnancy.
5.9 COC Use before or during Early Pregnancy
Extensive epidemiologic studies have revealed no increased risk of birth defects in women who have used oral contraceptives prior to pregnancy. Studies also do not suggest a teratogenic effect, particularly in so far as cardiac anomalies and limb reduction defects are concerned, when oral contraceptives are taken inadvertently during early pregnancy. Discontinue Finzala if pregnancy is confirmed.
Administration of oral contraceptives to induce withdrawal bleeding should not be used as a test for pregnancy [see Use in Specific Populations (8.1)].
5.10 Depression
Carefully observe women with a history of depression and discontinue Finzala if depression recurs to a serious degree.
5.11 Malignant Neoplasms
Breast Cancer
Finzala is contraindicated in females who currently have or have had breast cancer because breast cancer may be hormonally sensitive [see Contraindications (4)].
Epidemiology studies have not found a consistent association between use of combined oral contraceptives (COCs) and breast cancer risk. Studies do not show an association between ever (current or past) use of COCs and risk of breast cancer. However, some studies report a small increase in the risk of breast cancer among current or recent users (<6 months since last use) and current users with longer duration of COC use [see Adverse Reactions (6.2)].
Cervical Cancer
Some studies suggest that COCs are associated with an increase in the risk of cervical cancer or intraepithelial neoplasia. However, there is controversy about the extent to which these findings may be due to differences in sexual behavior and other factors.
5.12 Effect on Binding Globulins
The estrogen component of COCs may raise the serum concentrations of thyroxine-binding globulin, sex hormone-binding globulin, and cortisol-binding globulin. The dose of replacement thyroid hormones or cortisol therapy may need to be increased.
5.13 Monitoring
A woman who is taking COCs should have a yearly visit with her healthcare provider for a blood pressure check and for other indicated healthcare.
5.14 Hereditary Angioedema
In women with hereditary angioedema, exogenous estrogens may induce or exacerbate symptoms of angioedema.
5.15 Chloasma
Chloasma may occasionally occur, especially in women with a history of chloasma gravidarum. Women with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation while taking Finzala.
6. Adverse Reactions
The following serious adverse reactions with the use of COCs are discussed elsewhere in the labeling:
- Serious cardiovascular events and stroke [see Boxed Warning and Warnings and Precautions (5.1)]
- Vascular events [see Warnings and Precautions (5.1)]
- Liver disease [see Warnings and Precautions (5.2)]
Adverse reactions commonly reported by COC users are:
- Irregular uterine bleeding
- Nausea
- Breast tenderness
- Headache
6.1. Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to the rates in the clinical trials of another drug and may not reflect the rates observed in practice.
The data presented in Section 6.1 are from a clinical trial conducted with a 24-day regimen of norethindrone acetate 1 mg/ethinyl estradiol 20 mcg tablets. Norethindrone acetate and ethinyl estradiol tablets are bioequivalent to these norethindrone acetate/ethinyl estradiol tablets.
Common Adverse Reactions (Greater Than or Equal to 2% of all Treated Subjects): The most common adverse reactions reported by at least 2% of the 743 women using norethindrone acetate/ethinyl estradiol tablets were the following, in order of decreasing incidence: headache (6.3%), vaginal candidiasis (6.1%), nausea (4.6%), menstrual cramps (4.4%), breast tenderness (3.4%), bacterial vaginitis (3.1%), abnormal cervical smear (3.1%), acne (2.7%), mood swings (2.2%), and weight gain (2.0%).
Adverse Reactions Leading to Study Discontinuation: Among the 743 women using norethindrone acetate/ethinyl estradiol tablets, 46 women (6.2%) withdrew because of an adverse event. Adverse events occurring in 3 or more subjects leading to discontinuation of treatment were, in decreasing order: abnormal or irregular bleeding (1.3%), nausea (0.8%), menstrual cramps (0.5%), and increased blood pressure (0.4%).
6.2. Postmarketing Experience
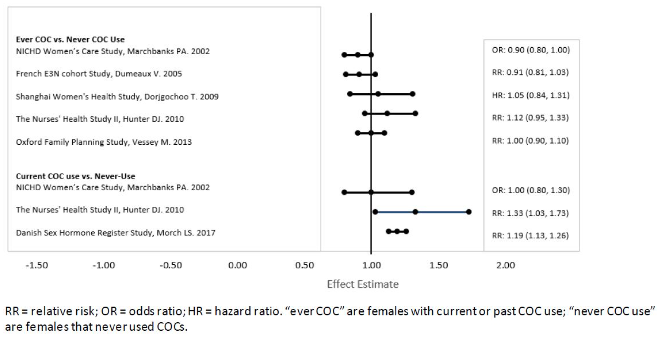
Five studies that compared breast cancer risk between ever-users (current or past use) of COCs and never-users of COCs reported no association between ever use of COCs and breast cancer risk, with effect estimates ranging from 0.90 to 1.12 (Figure 1).
Three studies compared breast cancer risk between current or recent COC users (<6 months since last use) and never users of COCs (Figure 1). One of these studies reported no association between breast cancer risk and COC use. The other two studies found an increased relative risk of 1.19 to 1.33 with current or recent use. Both of these studies found an increased risk of breast cancer with current use of longer duration, with relative risks ranging from 1.03 with less than one year of COC use to approximately 1.4 with more than 8 to 10 years of COC use.
Figure 1. Relevant Studies of Risk of Breast Cancer with Combined Oral Contraceptives:
The following adverse reactions have been identified during post approval use of a 24-day regimen of norethindrone acetate 1 mg/ethinyl estradiol 20 mcg tablets. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or evaluate a causal relationship to drug exposure.
Adverse reactions are grouped into System Organ Classes.
Vascular disorders: thrombosis/embolism (coronary artery, pulmonary, cerebral, deep vein).
Hepatobiliary disorders: cholelithiasis, cholecystitis, hepatic adenoma, hemangioma of liver.
Immune system disorders: hypersensitivity reaction.
Skin and subcutaneous disorders: alopecia, rash (generalized and allergic), pruritus, skin discoloration.
GI disorders: nausea, vomiting, abdominal pain.
Musculoskeletal and connective tissue disorders: myalgia.
Eye disorders: blurred vision, visual impairment, corneal thinning, change in corneal curvature (steepening).
Infections and infestations: fungal infection, vaginal infection.
Investigations: change in weight or appetite (increase or decrease), fatigue, malaise, peripheral edema, blood pressure increased.
Nervous system disorders: headache, dizziness, migraine, loss of consciousness.
Psychiatric disorders: mood swings, depression, insomnia, anxiety, suicidal ideation, panic attack, changes in libido.
Renal and urinary disorders: cystitis-like syndrome.
Reproductive system and breast disorders: breast changes (tenderness, pain, enlargement, and secretion), premenstrual syndrome, dysmenorrhea.
Cardiovascular: chest pain, palpitations, tachycardia, myocardial infarction.
7. Drug Interactions
Consult the labeling of the concurrently-used drug to obtain further information about interactions with COCs or the potential for enzyme alterations.
No drug-drug interaction studies were conducted with Finzala.
7.1 Effects of Other Drugs on Combined Oral Contraceptives
Substances diminishing the efficacy of COCs: Drugs or herbal products that induce certain enzymes, including cytochrome P450 3A4 (CYP3A4), may decrease the effectiveness of COCs or increase breakthrough bleeding. Some drugs or herbal products that may decrease the effectiveness of hormonal contraceptives include phenytoin, barbiturates, carbamazepine, bosentan, felbamate, griseofulvin, oxcarbazepine, rifampicin, topiramate and products containing St. John’s wort. Interactions between oral contraceptives and other drugs may lead to breakthrough bleeding and/or contraceptive failure. Counsel women to use an alternative method of contraception or a back-up method when enzyme inducers are used with COCs, and to continue back-up contraception for 28 days after discontinuing the enzyme inducer to ensure contraceptive reliability.
Substances increasing the plasma concentrations of COCs: Coadministration of atorvastatin and certain COCs containing ethinyl estradiol increase AUC values for ethinyl estradiol by approximately 20%. Ascorbic acid and acetaminophen may increase plasma ethinyl estradiol concentrations, possibly by inhibition of conjugation. CYP3A4 inhibitors such as itraconazole or ketoconazole may increase plasma hormone concentrations.
Human immunodeficiency virus (HIV)/Hepatitis C virus (HCV) protease inhibitors and non-nucleoside reverse transcriptase inhibitors: Significant changes (increase or decrease) in the plasma concentrations of the estrogen and progestin have been noted in some cases of coadministration of HIV/HCV protease inhibitors or of non-nucleoside reverse transcriptase inhibitors.
Antibiotics: There have been reports of pregnancy while taking hormonal contraceptives and antibiotics, but clinical pharmacokinetic studies have not shown consistent effects of antibiotics on plasma concentrations of synthetic steroids.
7.2 Effects of Combined Oral Contraceptives on Other Drugs
COCs containing ethinyl estradiol may inhibit the metabolism of other compounds. COCs have been shown to significantly decrease plasma concentrations of lamotrigine, likely due to induction of lamotrigine glucuronidation. This may reduce seizure control; therefore, dosage adjustments of lamotrigine may be necessary.
Women on thyroid hormone replacement therapy may need increased doses of thyroid hormone because serum concentration of thyroid-binding globulin increases with use of COCs.
7.3 Concomitant Use with HCV Combination Therapy – Liver Enzyme Elevation
Do not coadminister Finzala with HCV drug combinations containing ombitasvir/paritaprevir/ritonavir, with or without dasabuvir, due to potential for ALT elevations [see Warnings and Precautions (5.3)].
7.4 Interference with Laboratory Tests
The use of contraceptive steroids may influence the results of certain laboratory tests, such as coagulation factors, lipids, glucose tolerance, and binding proteins.
8.1. Pregnancy
Risk Summary
There is no use for contraception in pregnancy; therefore, Finzala should be discontinued during pregnancy. Epidemiologic studies and meta-analyses have not found an increased risk of genital or non-genital birth defects (including cardiac anomalies and limb-reduction defects) following exposure to COCs before conception or during early pregnancy.
In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2 to 4 percent and 15 to 20 percent, respectively.
8.2. Lactation
8.2 Lactation
Risk Summary
Contraceptive hormones and/or metabolites are present in human milk. COCs can reduce milk production in breastfeeding females. This reduction can occur at any time but is less likely to occur once breastfeeding is well-established. When possible, advise the nursing female to use other methods of contraception until she discontinues breastfeeding [see Dosage and Administration (2.2)]. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for Finzala and any potential adverse effects on the breastfed child from Finzala or from the underlying maternal condition.
8.4. Pediatric Use
Safety and efficacy of Finzala have been established in women of reproductive age. Efficacy is expected to be the same in postpubertal adolescents under the age of 18 years as for users 18 years and older. Use of this product before menarche is not indicated.
8.5. Geriatric Use
Finzala has not been studied in postmenopausal women and is not indicated in this population.
8.6. Renal Impairment
The pharmacokinetics of Finzala has not been studied in subjects with renal impairment.
8.7. Hepatic Impairment
The pharmacokinetics of Finzala has not been studied in subjects with hepatic impairment. However, steroid hormones may be poorly metabolized in patients with hepatic impairment. Acute or chronic disturbances of liver function may necessitate the discontinuation of COC use until markers of liver function return to normal and COC causation has been excluded [see Contraindications (4) and Warnings and Precautions (5.2)].
8.8 Body Mass Index
The safety and efficacy of Finzala in women with a body mass index (BMI) greater than 35 kg/m2 has not been evaluated [see Clinical Studies (14)].
© All content on this website, including data entry, data processing, decision support tools, "RxReasoner" logo and graphics, is the intellectual property of RxReasoner and is protected by copyright laws. Unauthorized reproduction or distribution of any part of this content without explicit written permission from RxReasoner is strictly prohibited. Any third-party content used on this site is acknowledged and utilized under fair use principles.